Chapter 19: Q86E (page 648)
The proton and carbon NMR spectra for each of three isomeric ketones with the formula C7H14Oare shown below. Assign a structure to each pair of spectra.
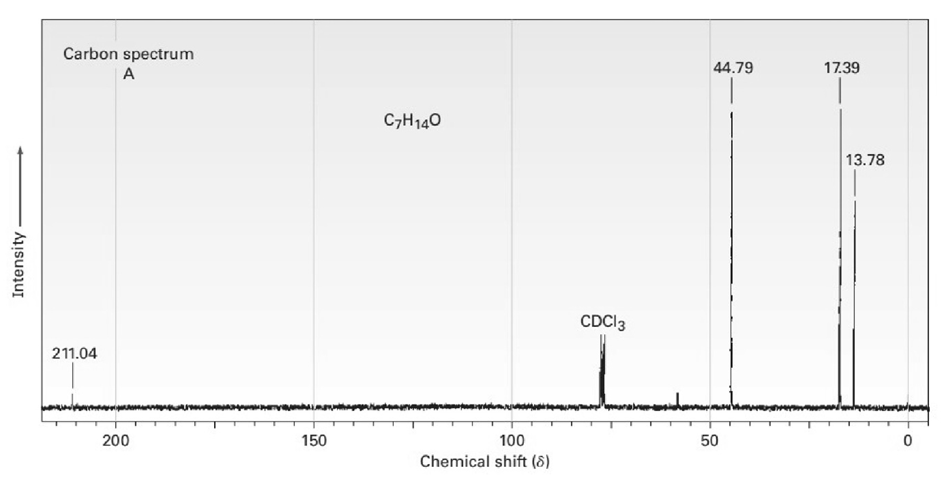
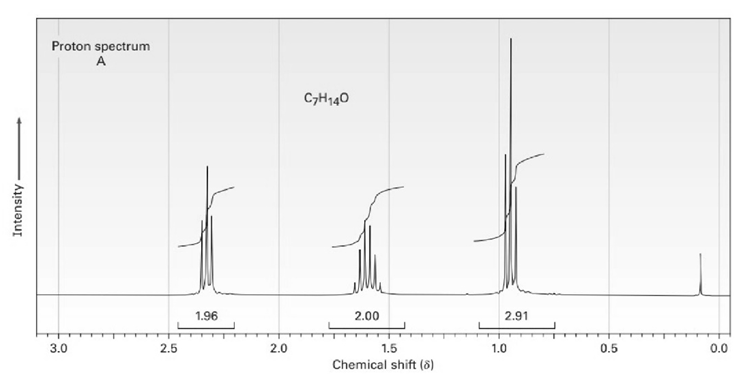

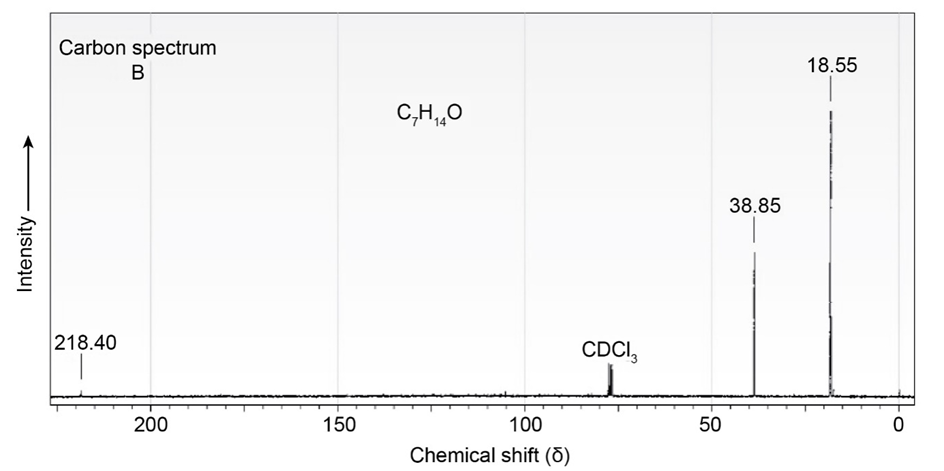
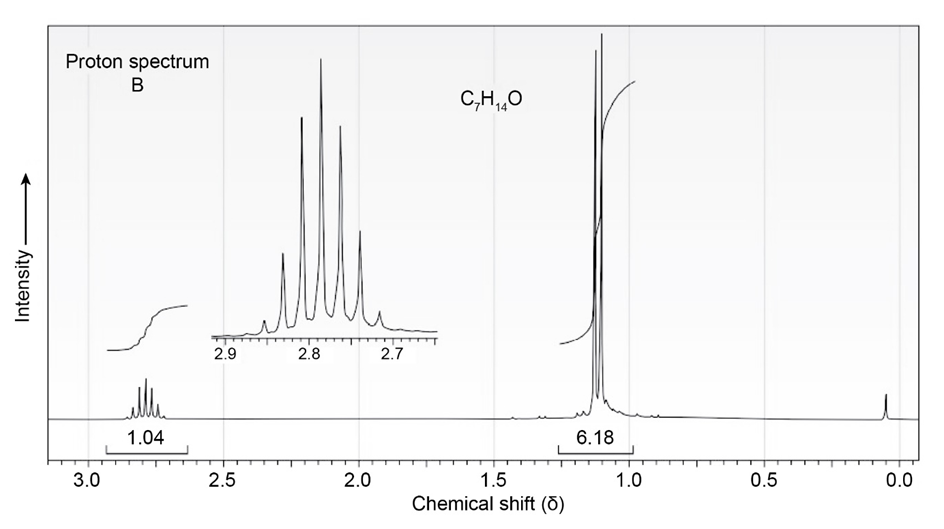
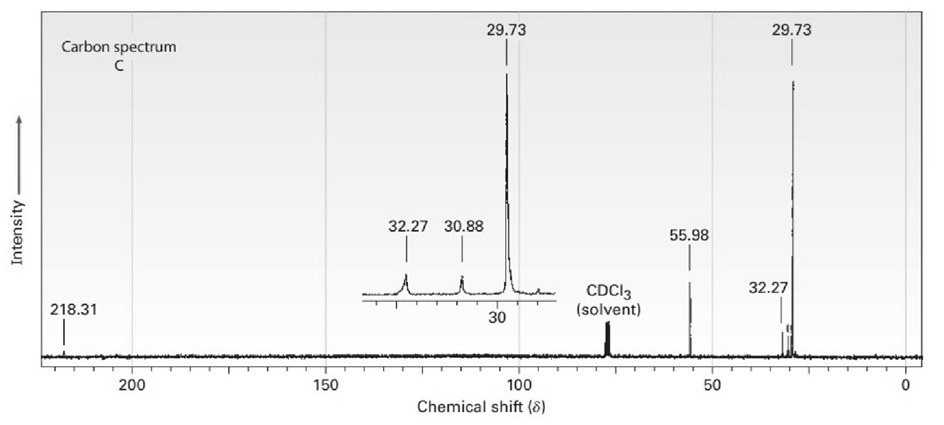

Short Answer
A)
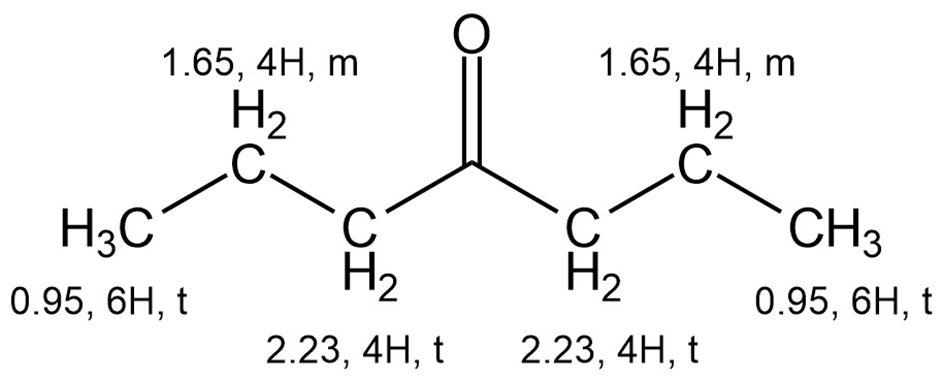
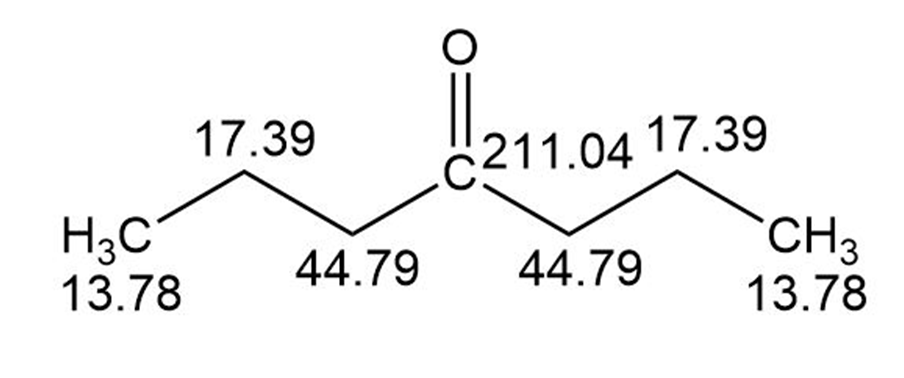
B)
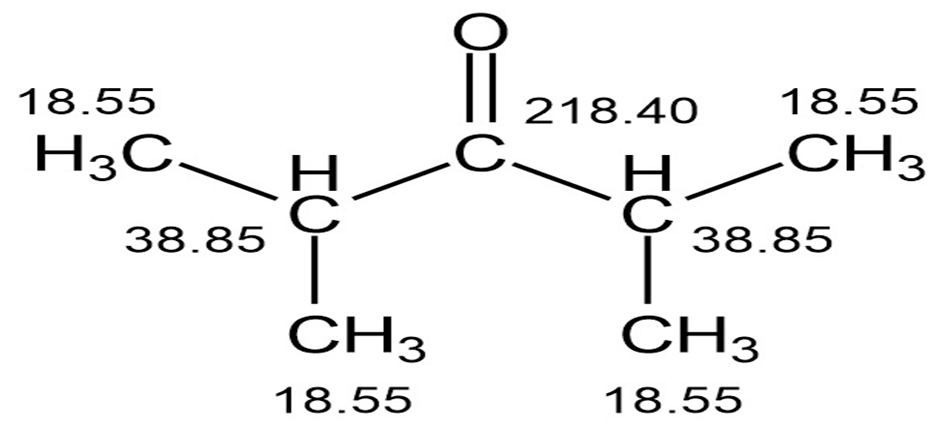
Assignment of the NMR spectrum
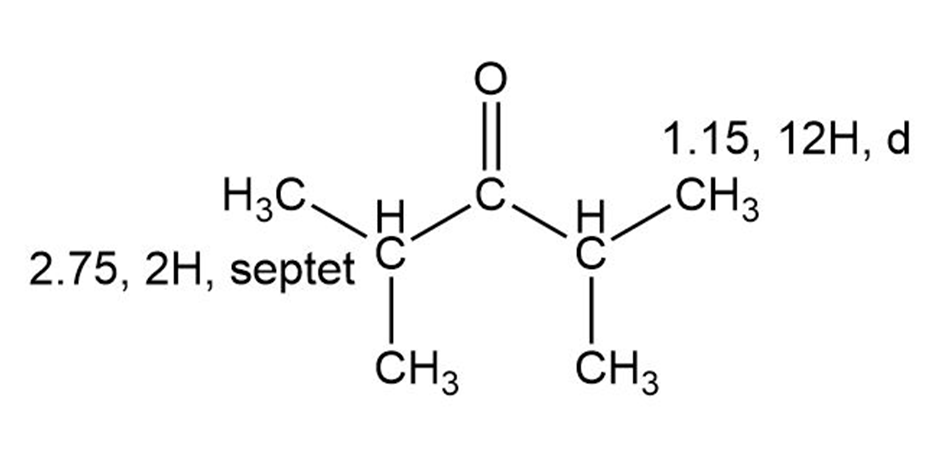
C)

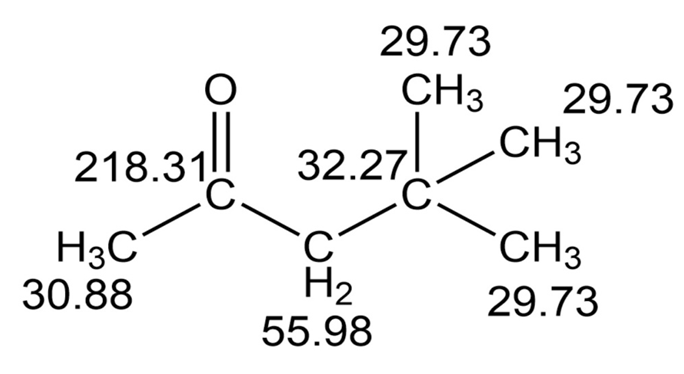
Learning Materials
Features
Discover
Chapter 19: Q86E (page 648)
The proton and carbon NMR spectra for each of three isomeric ketones with the formula C7H14Oare shown below. Assign a structure to each pair of spectra.







A)


B)


Assignment of the NMR spectrum
C)


All the tools & learning materials you need for study success - in one app.
Get started for free
Trans alkenes are converted into their cis isomers and vice versa on epoxidation followed by treatment of the epoxide with triphenylphosphine.Propose a mechanism for the reaction.
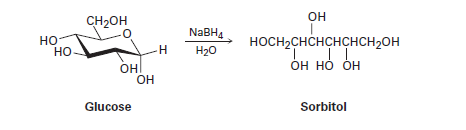
When glucose (Problem 19-52) is treated with\(NaB{H_4}\), reaction occurs to yield sorbitol, a polyalcohol commonly used as a food additive. Show how this reduction occurs.
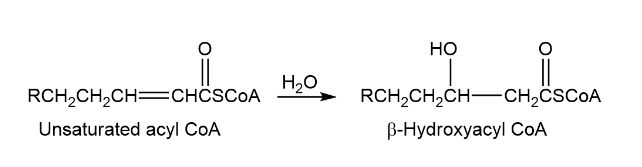
One of the steps in metabolism of fats is the reaction of an unsaturated acyl coA with water to give a - hydroxyacyl CoA. Propose a mechanism.
The following molecular model represents a tetrahedral intermediateresulting from addition of a nucleophile to an aldehyde or ketone. Identifythe reactants, and write the structure of the final product when thenucleophilic addition reaction is complete.
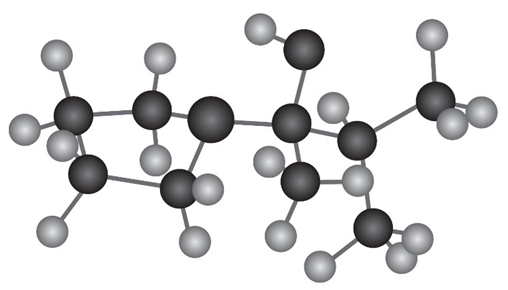
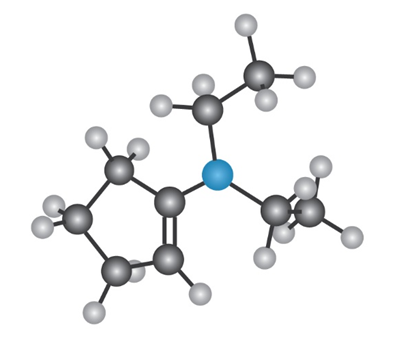
Draw the following molecule as a skeletal structure, and show how it can be prepared from a ketone and an amine.
What do you think about this solution?
We value your feedback to improve our textbook solutions.