Chapter 19: Q48E (page 648)
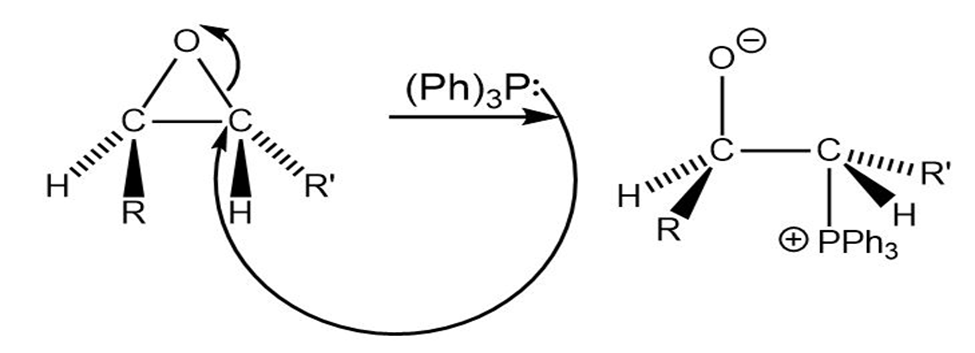
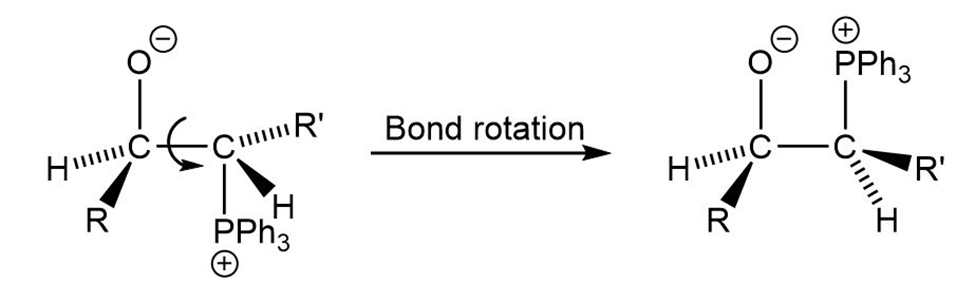
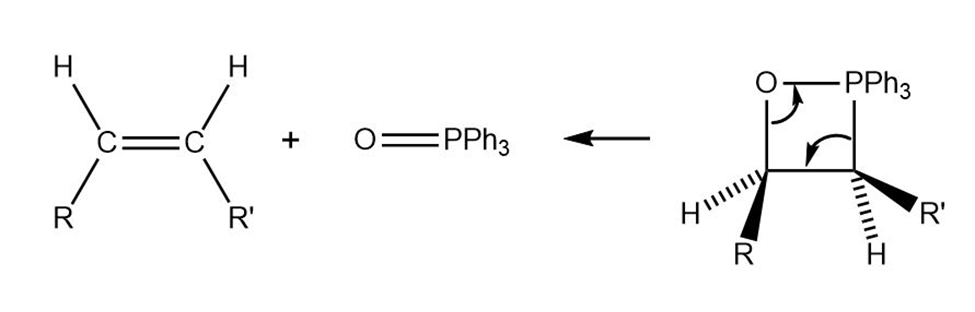
Trans alkenes are converted into their cis isomers and vice versa on epoxidation followed by treatment of the epoxide with triphenylphosphine.Propose a mechanism for the reaction.
Short Answer
Learning Materials
Features
Discover
Chapter 19: Q48E (page 648)
Trans alkenes are converted into their cis isomers and vice versa on epoxidation followed by treatment of the epoxide with triphenylphosphine.Propose a mechanism for the reaction.



All the tools & learning materials you need for study success - in one app.
Get started for free
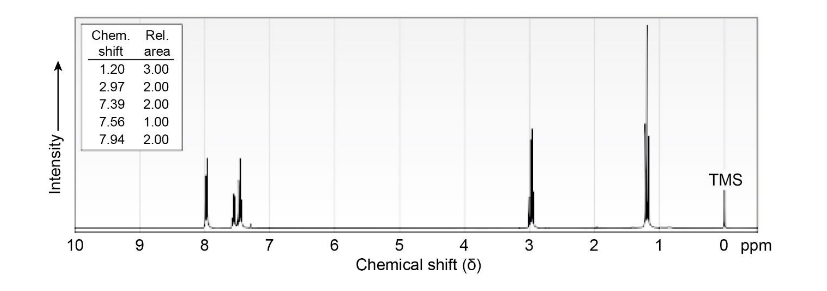
The 1HNMR spectrum shown is that of a compound with the formula C9H10O. How many double bonds and/or rings does this compound contain? If the unknown compound has an IR absorption at 1690 cm-1. , what is a likely structure?
The oxygen in water is primarily (99.8%) 16O, but water enriched with the heavy isotopeis also available. When an aldehyde or ketone is dissolvedin-enriched water, the isotopic label becomes incorporated into thecarbonyl group. Explain.
where O =
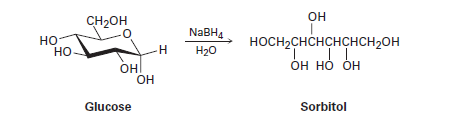
When glucose (Problem 19-52) is treated with\(NaB{H_4}\), reaction occurs to yield sorbitol, a polyalcohol commonly used as a food additive. Show how this reduction occurs.
Reaction of 2-butanone with HCN yields a chiral product. What stereochemistry does the product have? Is it optically active?
p-Nitrobenzaldehyde is more reactive toward nucleophilic additions than p-methoxybenzaldehyde. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.