Chapter 19: Q12P (page 624)
Draw the following molecule as a skeletal structure, and show how it can be prepared from a ketone and an amine.
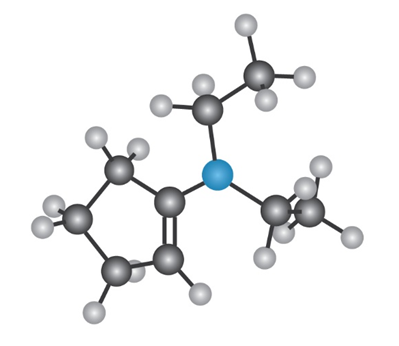
Short Answer
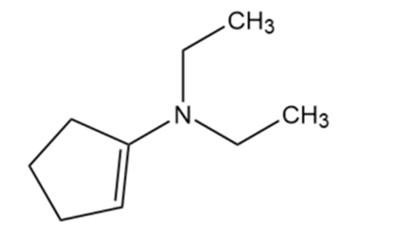
N, N-diethylcylopent-1en-1-amine
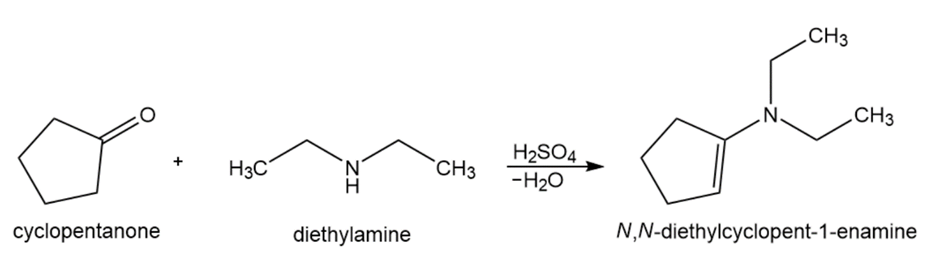
Formation of N, N-diethylcyclopent-1-enamine
Learning Materials
Features
Discover
Chapter 19: Q12P (page 624)
Draw the following molecule as a skeletal structure, and show how it can be prepared from a ketone and an amine.


N, N-diethylcylopent-1en-1-amine

Formation of N, N-diethylcyclopent-1-enamine
All the tools & learning materials you need for study success - in one app.
Get started for free
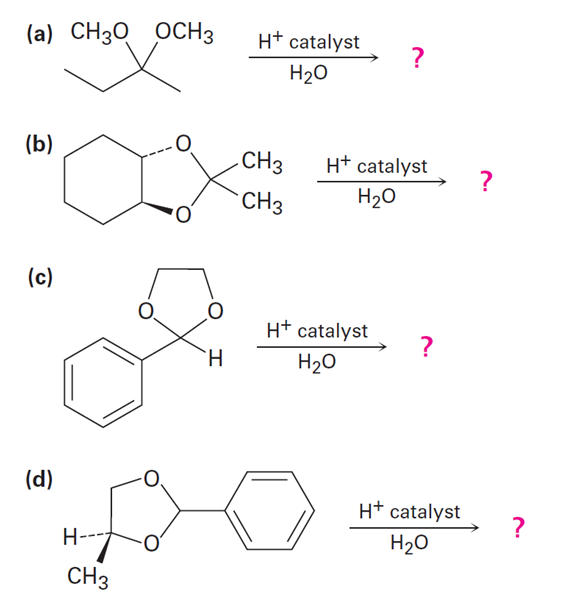
It is not uncommon for organic chemists to prepare acetals by an exchange-type process known as transacetalization. Predict the product(s) and show the mechanism for the transacetalization reactions below.
a.
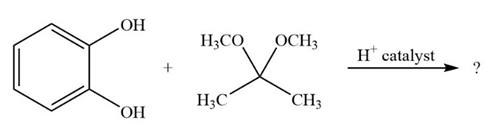
b.
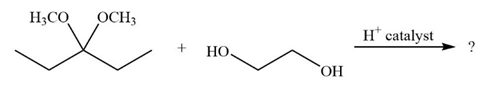
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?
How might you use mass spectrometry to distinguish between the following pairs of isomers?
(a) 3-Methyl-2-hexanone and 4-methyl-2-hexanone
(b) 3-Heptanone and 4-heptanone
(c) 2-Methylpentanal and 3-methylpentanal
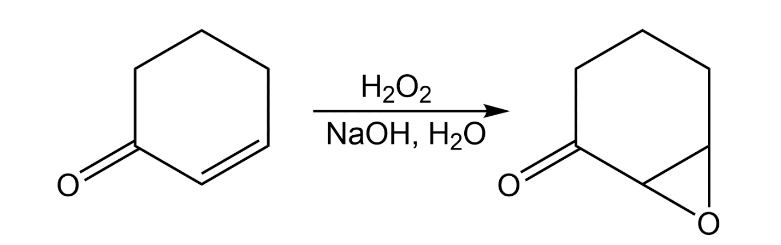
Treatment of an -unsaturated ketone with basic aqueous hydrogen peroxide yields an epoxy ketone. The reaction is specific to unsaturated ketones; isolated alkene double bonds do not react. Propose a mechanism.
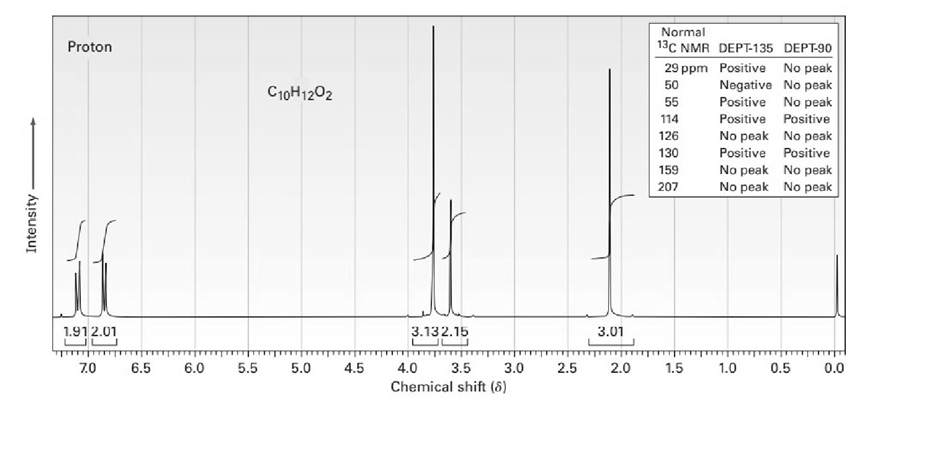
The proton NMR spectrum for a compound with formula C10H12O2is shown below. The infrared spectrum has a strong band at. The broadband-decoupled 13CNMR spectral results are tabulated along with the DEPT-135 and DEPT-90 information. Draw the structure of this compound.
What do you think about this solution?
We value your feedback to improve our textbook solutions.