Chapter 19: Q33E (page 648)
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?
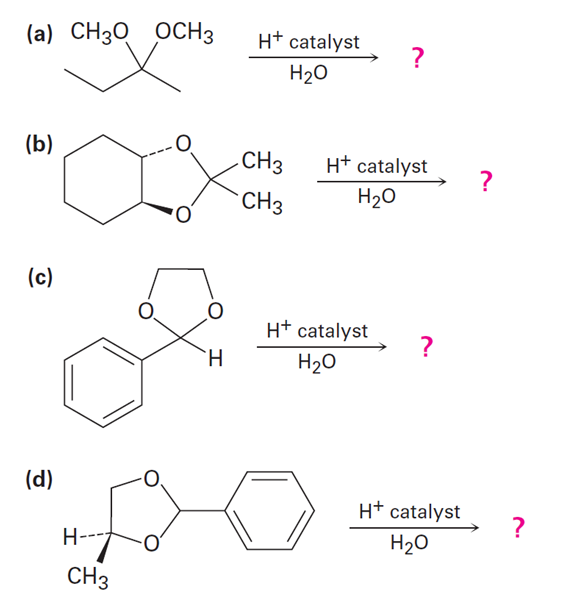
Short Answer
a.
b. No reaction possible.
c.
d.
Learning Materials
Features
Discover
Chapter 19: Q33E (page 648)
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?

a.

b. No reaction possible.
c.

d.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Predict the products of the wolff-Kishner reduction reactions below. Provide the electron pushing mechanism for each, beginning from the hydrazone intermediate.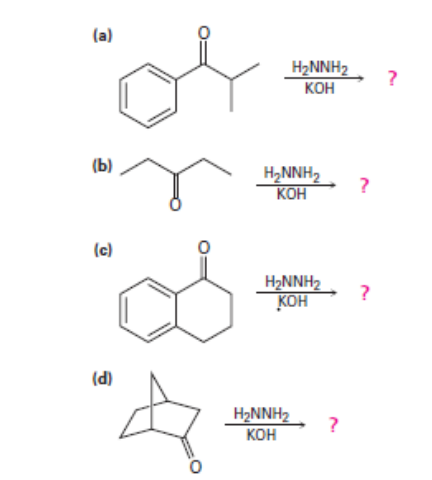
Treatment of an aldehyde or ketone with cyanide ion (), followed by protonation of the tetrahedral alkoxide ion intermediate, gives a cyanohydrin.
Show the structure of the cyanohydrin obtained from cyclohexanone.
Treatment of 2-cyclohexenone with HCN/KCN yields a saturated keto nitrile rather than an unsaturated cyanohydrin. Show the structure of the product,
and propose a mechanism for the reaction.
How might you use IR spectroscopy to determine whether reaction between2-cyclohexenoneand lithium dimethylcopper gives the direct addition productor the conjugate addition product?
The Wharton reaction converts an epoxy ketone to an allylic alcohol by reaction with hydrazine. Propose a mechanism. (Hint: Review the Wolff–Kishner reaction in Section 19-9.)
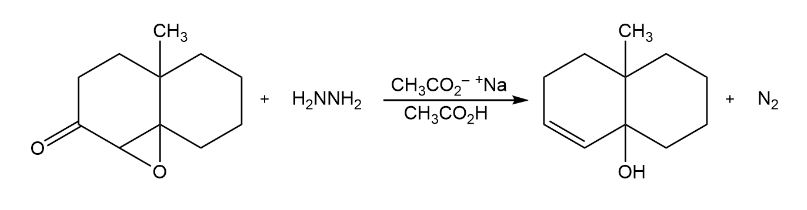
What do you think about this solution?
We value your feedback to improve our textbook solutions.