Chapter 19: Q31E (page 648)
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?
Short Answer
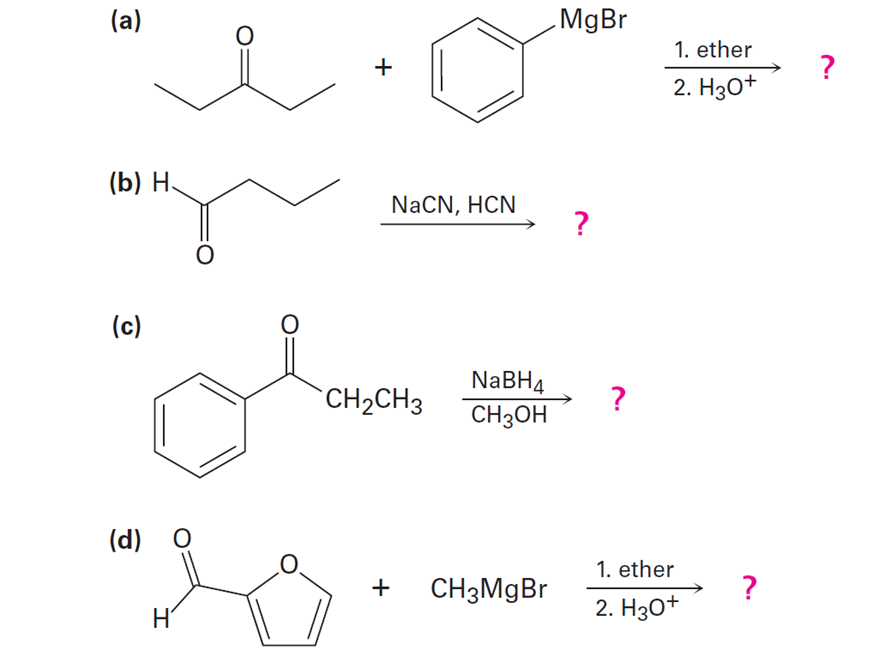
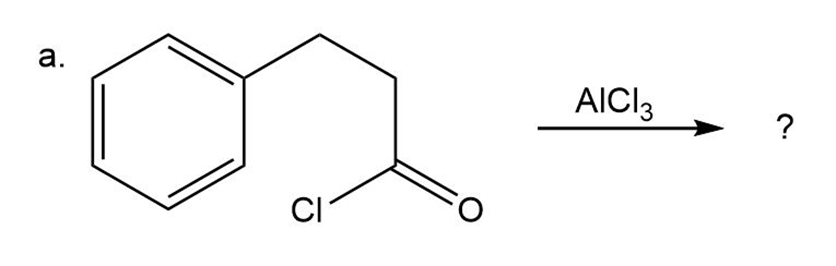
a)
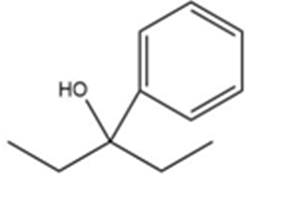
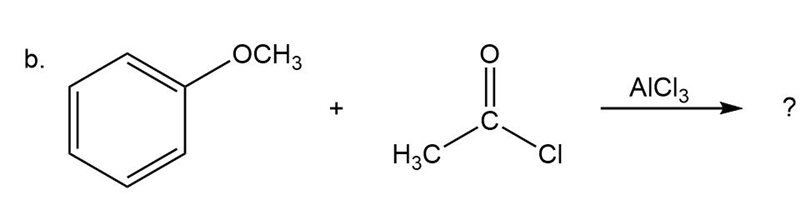
b)
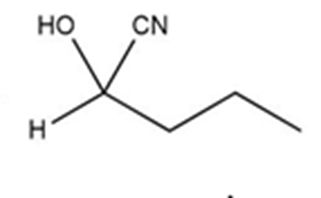
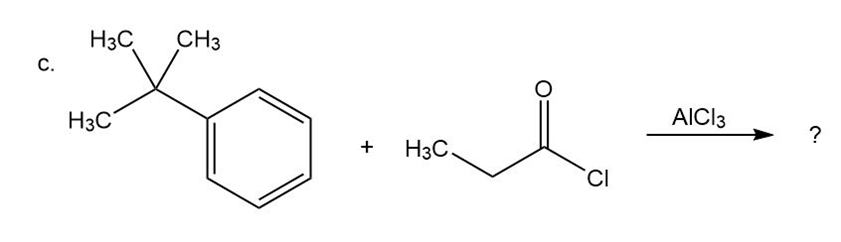
c)
d)
Learning Materials
Features
Discover
Chapter 19: Q31E (page 648)
Predict the product(s) and provide the mechanism for each reaction below. What does each mechanism have in common?

a)

b)

c)

d)

All the tools & learning materials you need for study success - in one app.
Get started for free
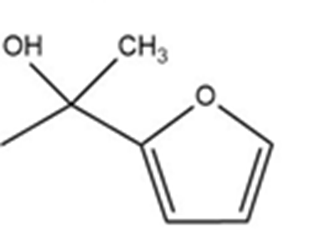
Predict the product(s) and provide the mechanism for each reactionbelow. What does each mechanism have in common?
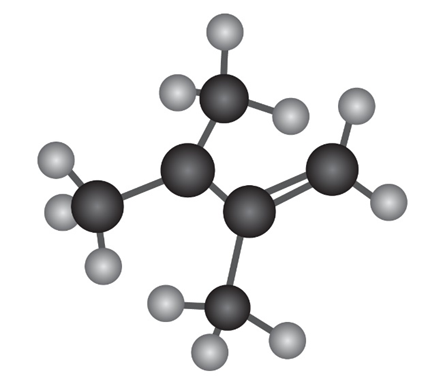
The enamine prepared from acetone and dimethylamine is shown in its
lowest-energy form.
(a) What is the geometry and hybridization of the nitrogen atom?
(b) What orbital on nitrogen holds the lone pair of electrons?
(c) What is the geometric relationship between the p orbitals of the
double bond and the nitrogen orbital that holds the lone pair? Why
do you think this geometry represents the minimum energy?
The oxygen in water is primarily (99.8%) 16O, but water enriched with the heavy isotopeis also available. When an aldehyde or ketone is dissolvedin-enriched water, the isotopic label becomes incorporated into thecarbonyl group. Explain.
where O =
How would you carry out the following reactions? More than one step may be
required.
(a) 3-Hexyne3-Hexanone
(b) Benzenem-Bromoacetophenone
(c) BromobenzeneAcetophenone
(d) 1-Methylcyclohexene2-Methylcyclohexanone
Question: Predict the products of the wolff-Kishner reduction reactions below. Provide the electron pushing mechanism for each, beginning from the hydrazone intermediate.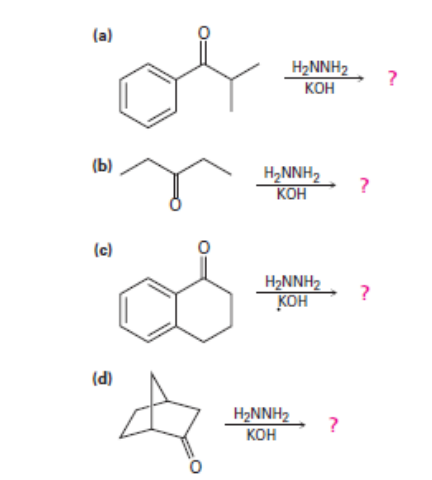
What do you think about this solution?
We value your feedback to improve our textbook solutions.