Chapter 19: Q40E (page 648)
One of the steps in metabolism of fats is the reaction of an unsaturated acyl coA with water to give a - hydroxyacyl CoA. Propose a mechanism.
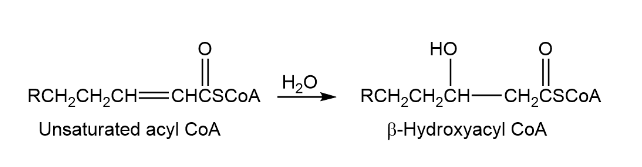
Short Answer
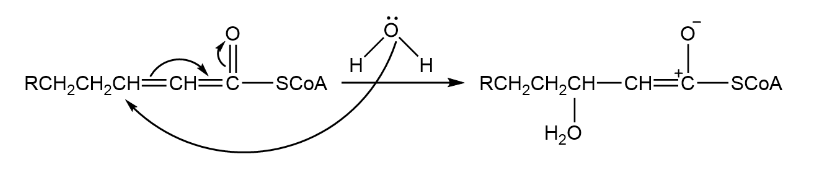
The first step of the mechanism is-
Learning Materials
Features
Discover
Chapter 19: Q40E (page 648)
One of the steps in metabolism of fats is the reaction of an unsaturated acyl coA with water to give a - hydroxyacyl CoA. Propose a mechanism.

The first step of the mechanism is-

All the tools & learning materials you need for study success - in one app.
Get started for free
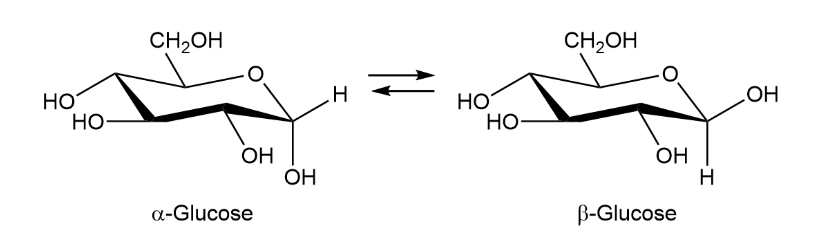
When crystals of pure -glucose are dissolved in water, isomerization occurs slowly to produce -glucose. Propose a mechanism for isomerization.
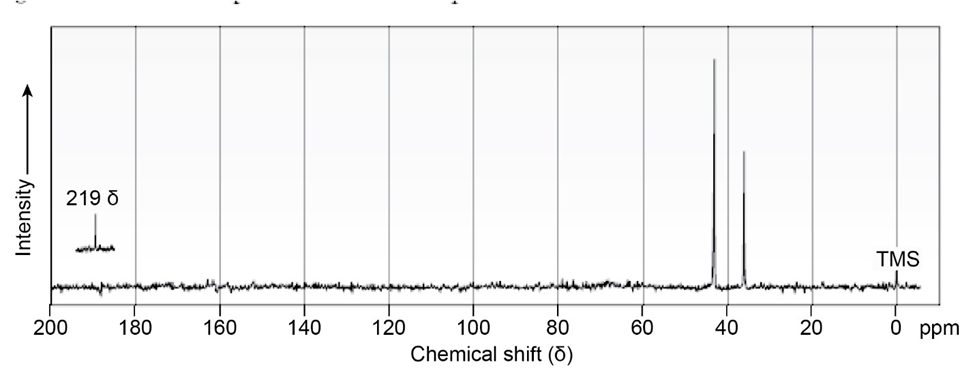
Compound A, , has an intense IR absorption at 1750 and gives the 13C NMR spectrum shown. Propose a structure for A.
Treatment of an aldehyde or ketone with cyanide ion (), followed by protonation of the tetrahedral alkoxide ion intermediate, gives a cyanohydrin.
Show the structure of the cyanohydrin obtained from cyclohexanone.
Question: Predict the products of the wolff-Kishner reduction reactions below. Provide the electron pushing mechanism for each, beginning from the hydrazone intermediate.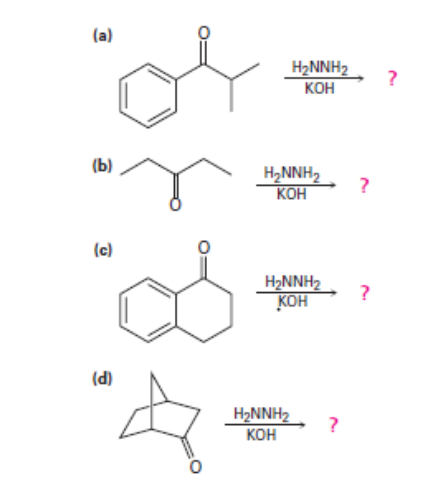
How would you carry out the following reactions? More than one step may be
required.
(a) 3-Hexyne3-Hexanone
(b) Benzenem-Bromoacetophenone
(c) BromobenzeneAcetophenone
(d) 1-Methylcyclohexene2-Methylcyclohexanone
What do you think about this solution?
We value your feedback to improve our textbook solutions.