Chapter 19: Q3P (page 608)
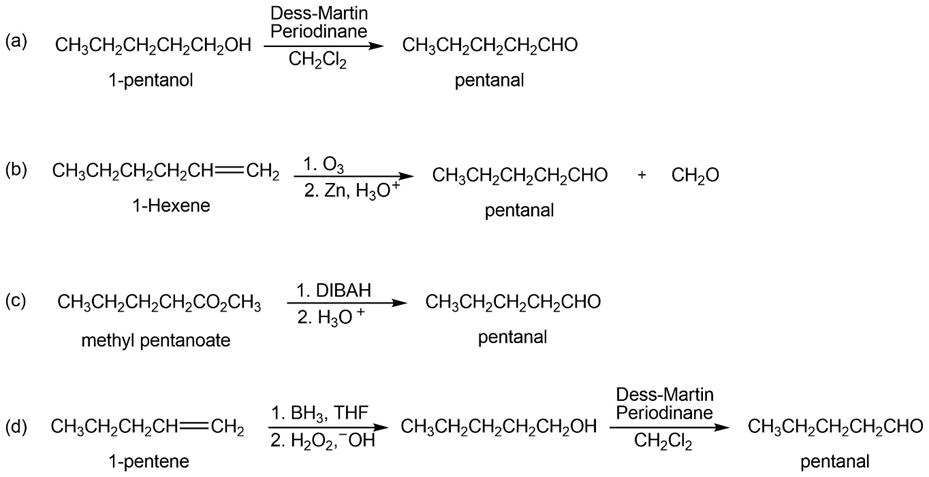
How would you prepare pentanal from the following starting materials?
(a) CH3CH2CH2CH2CH2OH (b) CH3CH2CH2CH2CH=CH2
(c) CH3CH2CH2CH2CO2CH3 (d) CH3CH2CH2CH=CH2
Short Answer
Learning Materials
Features
Discover
Chapter 19: Q3P (page 608)
How would you prepare pentanal from the following starting materials?
(a) CH3CH2CH2CH2CH2OH (b) CH3CH2CH2CH2CH=CH2
(c) CH3CH2CH2CH2CO2CH3 (d) CH3CH2CH2CH=CH2

All the tools & learning materials you need for study success - in one app.
Get started for free
Propose a mechanism to account for the formation of 3,5-dimethyl pyrazole from hydrazine and 2,4-pentanedione. Look carefully to see what has happened to each carbonyl carbon in going from starting material to product.
The SN2 reaction of (dibromomethyl)benzene, C6H5CHBr2, withNaOH yields benzaldehyde rather than (dihydroxymethyl)benzene,C6H5CH(OH)2. Explain.
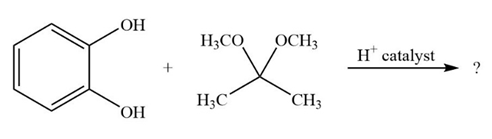
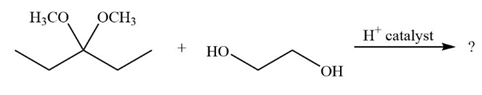
It is not uncommon for organic chemists to prepare acetals by an exchange-type process known as transacetalization. Predict the product(s) and show the mechanism for the transacetalization reactions below.
a.
b.
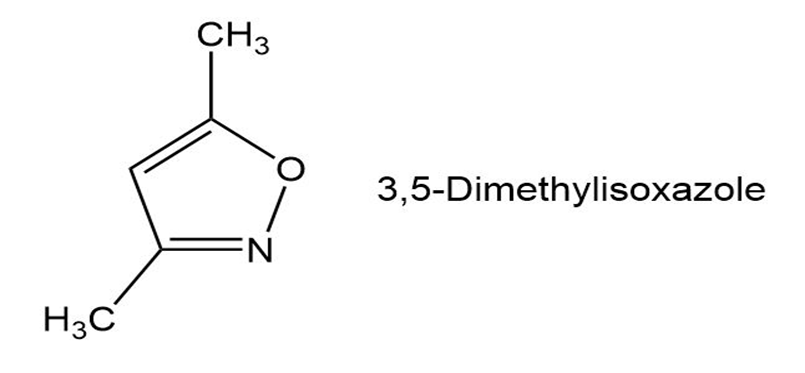
In light of your answer to Problem 19-46, propose a mechanism for the formation of 3, 5-dimethylisoxazole from hydroxylamine and 2,4-pentanedione
Show all the steps in the acid-catalyzed formation of a cyclic acetal from ethylene glycol and an aldehyde or ketone.
What do you think about this solution?
We value your feedback to improve our textbook solutions.