Chapter 19: Q42E (page 648)
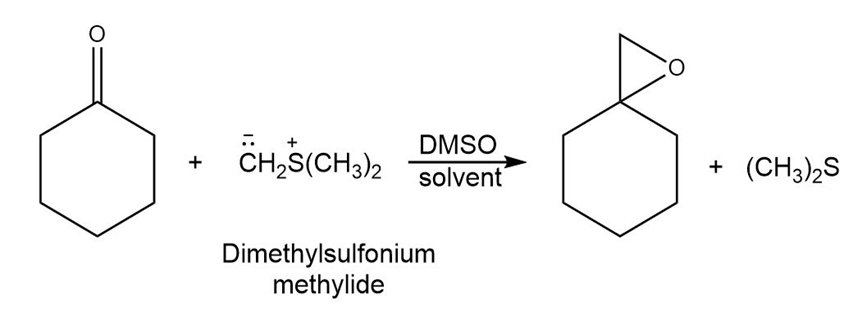
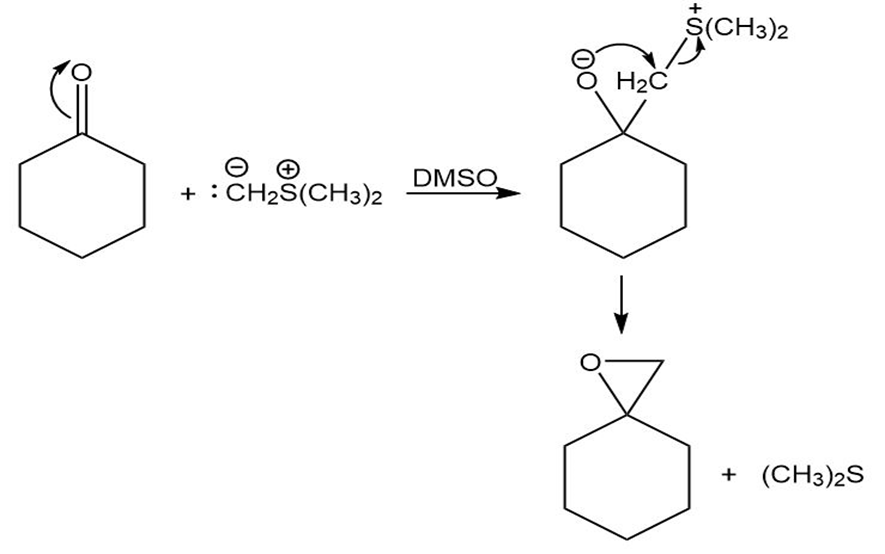
Ketones react with dimethylsulfonium methylide to yield epoxides. Suggest a mechanism for the reaction.
Short Answer
Learning Materials
Features
Discover
Chapter 19: Q42E (page 648)
Ketones react with dimethylsulfonium methylide to yield epoxides. Suggest a mechanism for the reaction.


All the tools & learning materials you need for study success - in one app.
Get started for free
Propose structures for ketones or aldehydes that have the following NMR spectra:
(a)C10H12O
localid="1656491160287"
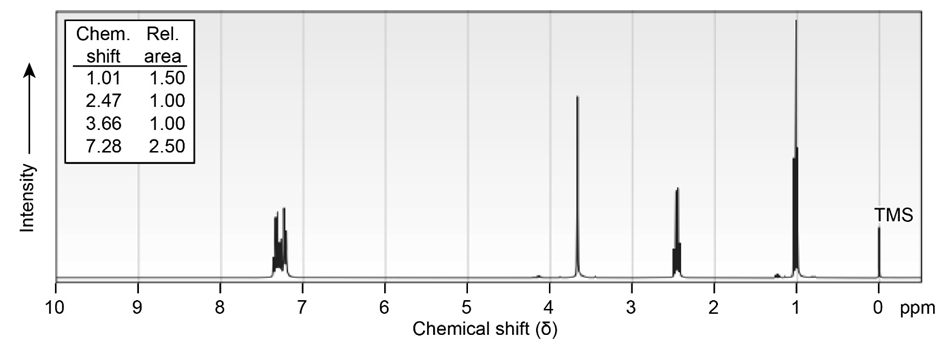
(b) localid="1656491164732"
localid="1656491168876"
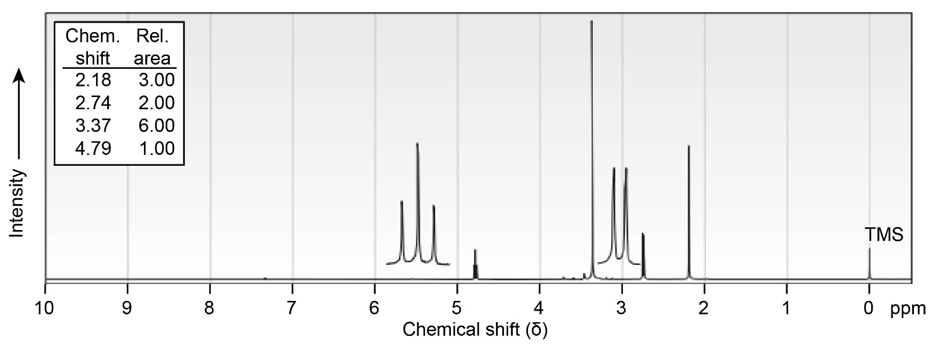
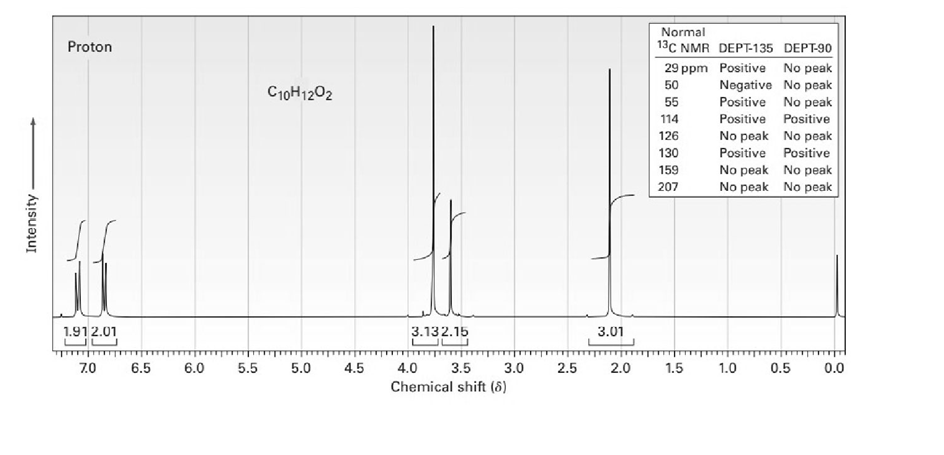
The proton NMR spectrum for a compound with formula C10H12O2is shown below. The infrared spectrum has a strong band at. The broadband-decoupled 13CNMR spectral results are tabulated along with the DEPT-135 and DEPT-90 information. Draw the structure of this compound.
Draw and name the seven aldehydes and ketones with the formula . Which are chiral?
The following molecular model represents a tetrahedral intermediateresulting from addition of a nucleophile to an aldehyde or ketone. Identifythe reactants, and write the structure of the final product when thenucleophilic addition reaction is complete.
p-Nitrobenzaldehyde is more reactive toward nucleophilic additions than p-methoxybenzaldehyde. Explain.
What do you think about this solution?
We value your feedback to improve our textbook solutions.