Chapter 19: Q84E (page 648)
Propose structures for ketones or aldehydes that have the following NMR spectra:
(a)C10H12O
localid="1656491160287"
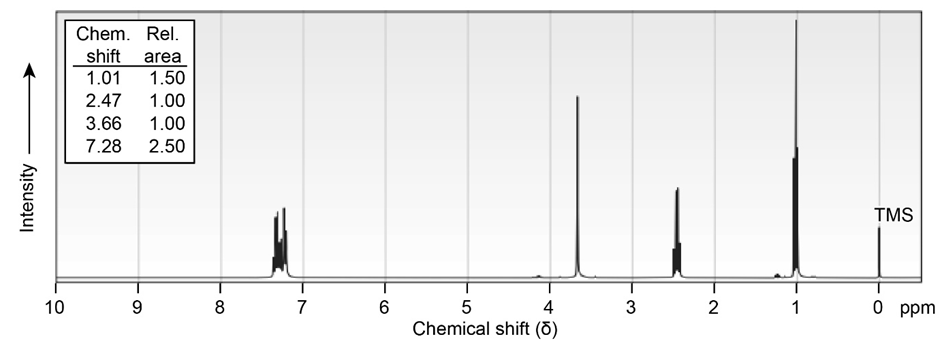
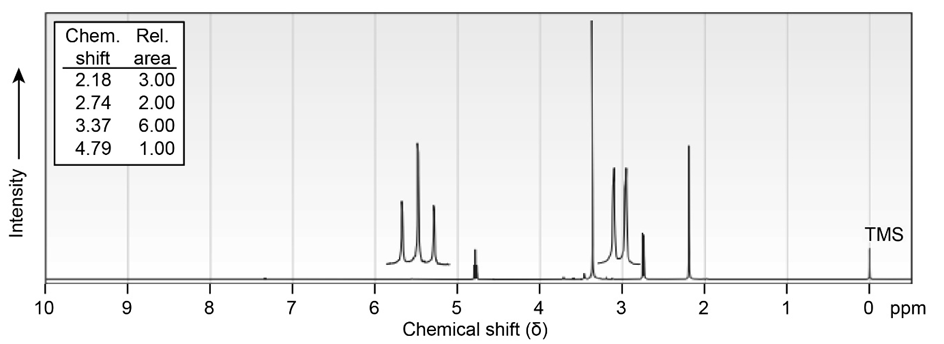
(b) localid="1656491164732"
localid="1656491168876"
Short Answer
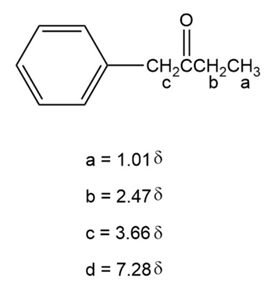
The structure of compound is:
a.
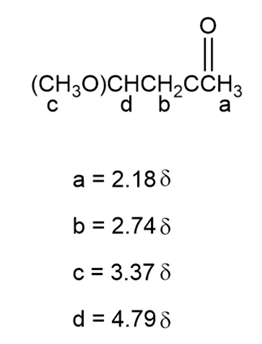
b
Learning Materials
Features
Discover
Chapter 19: Q84E (page 648)
Propose structures for ketones or aldehydes that have the following NMR spectra:
(a)C10H12O
localid="1656491160287"

(b) localid="1656491164732"
localid="1656491168876"

The structure of compound is:
a.

b

All the tools & learning materials you need for study success - in one app.
Get started for free
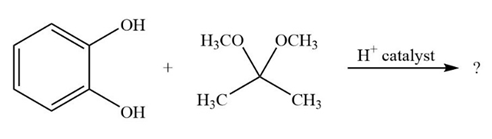
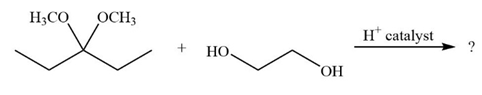
It is not uncommon for organic chemists to prepare acetals by an exchange-type process known as transacetalization. Predict the product(s) and show the mechanism for the transacetalization reactions below.
a.
b.
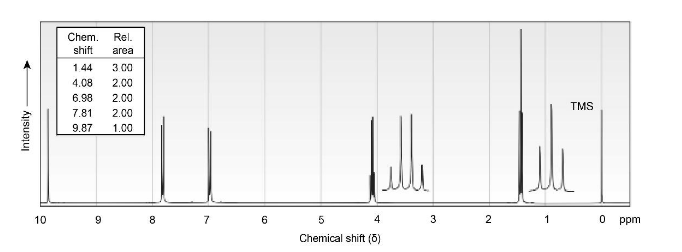
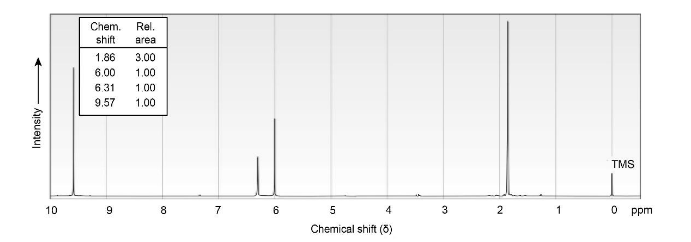
Propose structures for ketones or aldehydes that have the following 1H NMR spectra:
(a) C9H10O2
IR: 1695 cm-1
(b) C4H6O
IR: 1690 cm-1
Show all the steps in the acid-catalyzed formation of a cyclic acetal from ethylene glycol and an aldehyde or ketone.
How might you use mass spectrometry to distinguish between the following pairs of isomers?
(a) 3-Methyl-2-hexanone and 4-methyl-2-hexanone
(b) 3-Heptanone and 4-heptanone
(c) 2-Methylpentanal and 3-methylpentanal
Acid-catalyzed dehydration of 3-hydroxy-3-phenylcyclohexanone leads to an unsaturated ketone. What possible structures are there for the product? At what position in the IR spectrum would you expect each to absorb? If the actual product has an absorption at , what is its structure?
What do you think about this solution?
We value your feedback to improve our textbook solutions.