Chapter 19: Q65E (page 648)
How would you synthesize the following compounds from cyclohexanone?
- 1-Methylcyclohexene
- 2-Phenylcyclohexanone
- cis-1,2-Cyclohexanediol
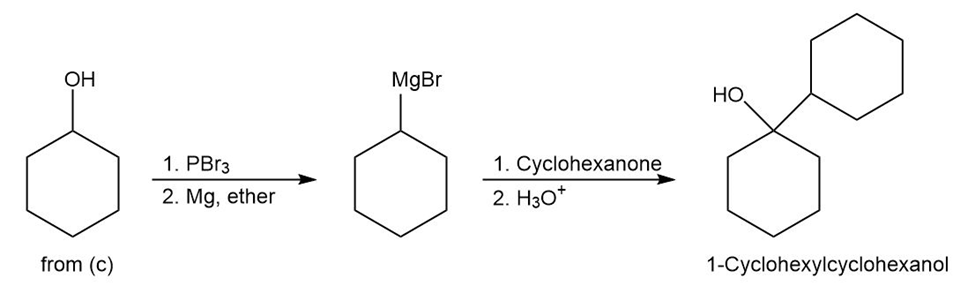
- 1-Cyclohexylcyclohexanol
Short Answer
a)
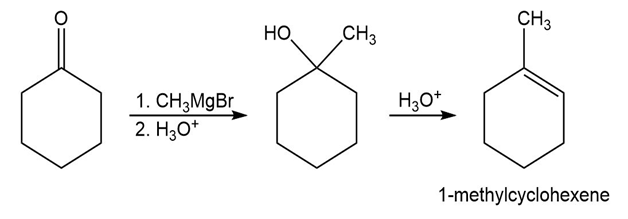
b)

c)

d)
Learning Materials
Features
Discover
Chapter 19: Q65E (page 648)
How would you synthesize the following compounds from cyclohexanone?
a)

b)

c)

d)

All the tools & learning materials you need for study success - in one app.
Get started for free
The Wharton reaction converts an epoxy ketone to an allylic alcohol by reaction with hydrazine. Propose a mechanism. (Hint: Review the Wolff–Kishner reaction in Section 19-9.)
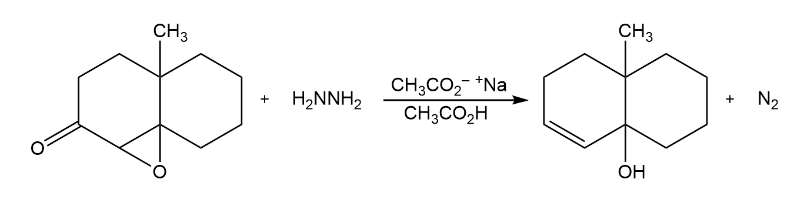
Acid-catalyzed dehydration of 3-hydroxy-3-phenylcyclohexanone leads to an unsaturated ketone. What possible structures are there for the product? At what position in the IR spectrum would you expect each to absorb? If the actual product has an absorption at , what is its structure?
How would you prepare pentanal from the following starting materials?
(a) CH3CH2CH2CH2CH2OH (b) CH3CH2CH2CH2CH=CH2
(c) CH3CH2CH2CH2CO2CH3 (d) CH3CH2CH2CH=CH2
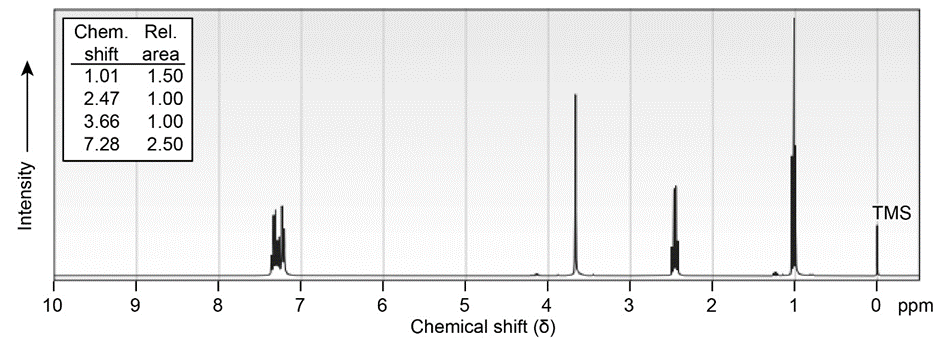
Propose structures for ketones or aldehydes that have the following \(^1H\;NMR\) spectra:
(a) \({C_{10}}{H_{12}}O\)
IR: 1710 \(c{m^{ - 1}}\)
(b)\({C_6}{H_{12}}{O_3}\)
IR: 1715\(c{m^{ - 1}}\)
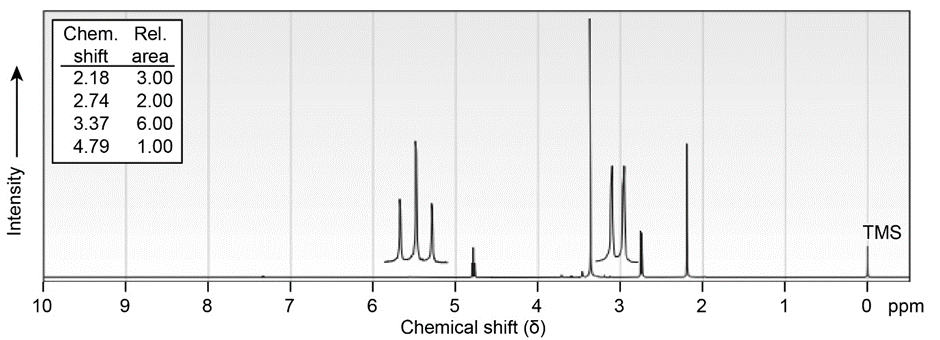
In light of your answer to Problem 19-46, propose a mechanism for the formation of 3, 5-dimethylisoxazole from hydroxylamine and 2,4-pentanedione
What do you think about this solution?
We value your feedback to improve our textbook solutions.