Chapter 19: Q45E (page 648)
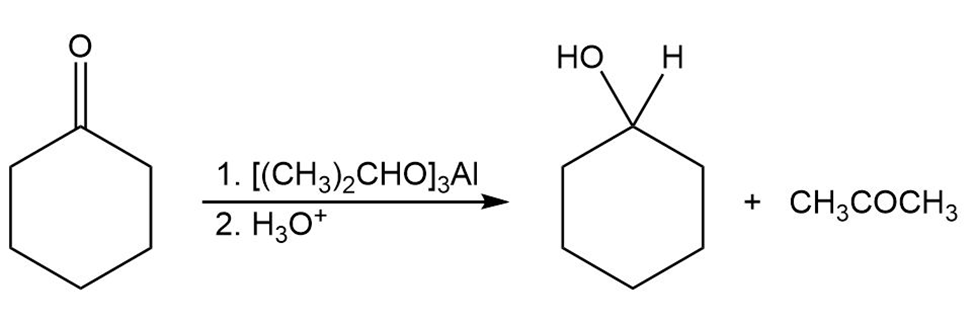
The Meerwein–Ponndorf–Verley reaction involves the reduction of a ketone by treatment with an excess of aluminum triisopropoxide,.The mechanism of the process is closely related to the Cannizzaro reaction in that a hydride ion acts as a leaving group. Propose a mechanism.
Short Answer
 Formation of Complex
Formation of Complex
 Formation of Alcohol
Formation of Alcohol
 Formation of Complex
Formation of Complex Formation of Alcohol
Formation of Alcohol