Chapter 19: Aldehydes and Ketones: Nucleophilic Addition Reactions
Q43E
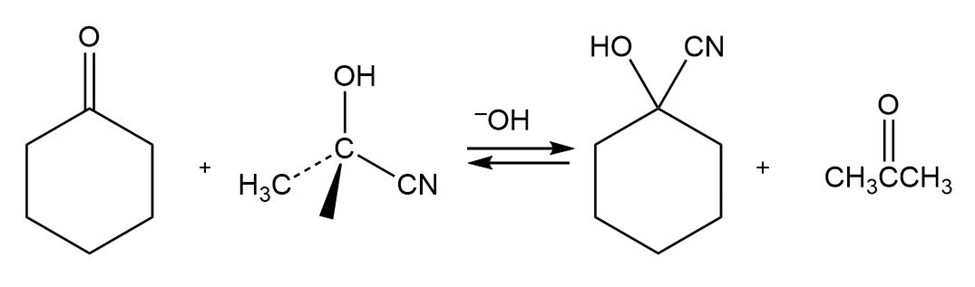
When cyclohexanone is heated in the presence of a large amount of acetone cyanohydrin and a small amount of base, cyclohexanone cyanohydrin and acetone are formed. Propose a mechanism.
Q44E
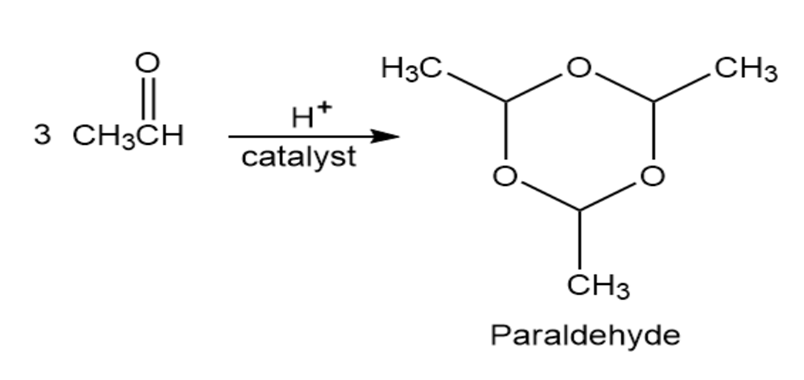
Paraldehyde, a sedative, and hypnotic agent, is prepared by treatment of acetaldehyde with an acidic catalyst. Propose a mechanism for the reaction.
Q45E
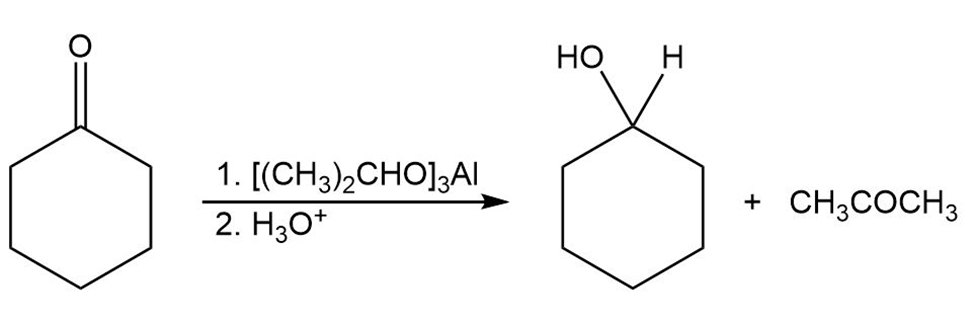
The Meerwein–Ponndorf–Verley reaction involves the reduction of a ketone by treatment with an excess of aluminum triisopropoxide,.The mechanism of the process is closely related to the Cannizzaro reaction in that a hydride ion acts as a leaving group. Propose a mechanism.
Q46E
Propose a mechanism to account for the formation of 3,5-dimethyl pyrazole from hydrazine and 2,4-pentanedione. Look carefully to see what has happened to each carbonyl carbon in going from starting material to product.
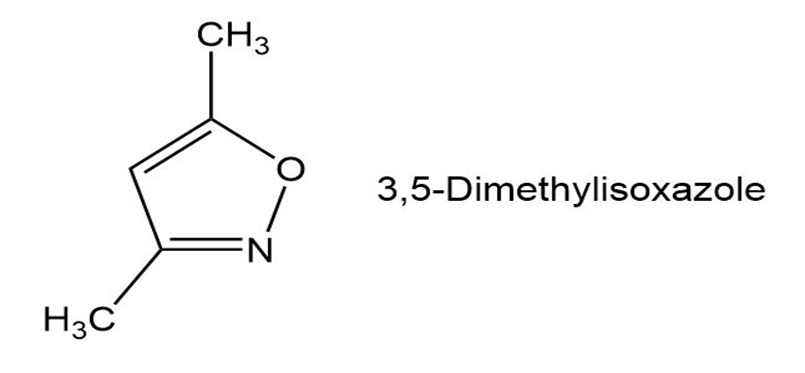
Q47E
In light of your answer to Problem 19-46, propose a mechanism for the formation of 3, 5-dimethylisoxazole from hydroxylamine and 2,4-pentanedione
Q48E
Trans alkenes are converted into their cis isomers and vice versa on epoxidation followed by treatment of the epoxide with triphenylphosphine.Propose a mechanism for the reaction.
Q49E
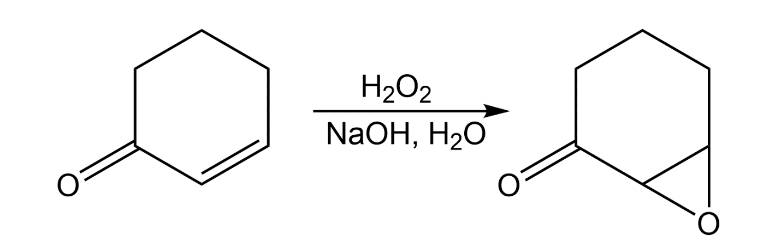
Treatment of an -unsaturated ketone with basic aqueous hydrogen peroxide yields an epoxy ketone. The reaction is specific to unsaturated ketones; isolated alkene double bonds do not react. Propose a mechanism.
Q4P
How would you carry out the following reactions? More than one step may be
required.
(a) 3-Hexyne3-Hexanone
(b) Benzenem-Bromoacetophenone
(c) BromobenzeneAcetophenone
(d) 1-Methylcyclohexene2-Methylcyclohexanone
Q51E
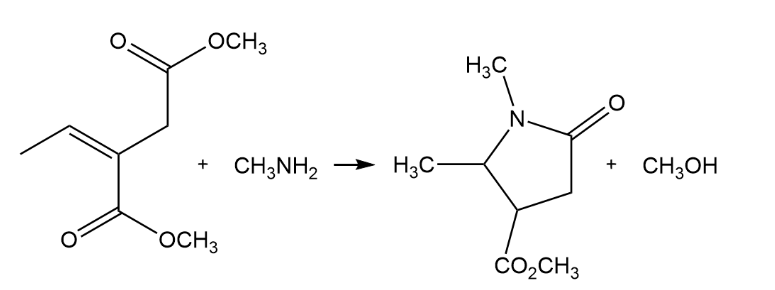
Primary amines react with esters to yield amides: RCO2R' + R"NH2 RCONHR" + R'OH . Propose a mechanism for the following reaction of an a b-unsaturated ester.
Q52E
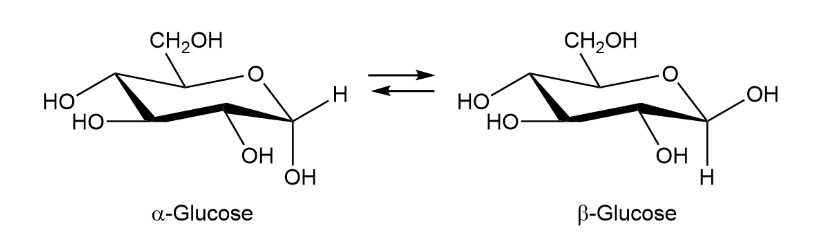
When crystals of pure -glucose are dissolved in water, isomerization occurs slowly to produce -glucose. Propose a mechanism for isomerization.