Chapter 1: Q2. (page 12)
Question: Label each bond in the following compounds as ionic or covalent.
Short Answer
Answer
:Covalent
Ionic
role="math" localid="1648114228071" : Covalent
:Both covalent and ionic
:Both covalent and ionic
Learning Materials
Features
Discover
Chapter 1: Q2. (page 12)
Question: Label each bond in the following compounds as ionic or covalent.
Answer
:Covalent
Ionic
role="math" localid="1648114228071" : Covalent
:Both covalent and ionic
:Both covalent and ionic
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Benzene is the simplest member of a whole class of compounds called aromatic hydrocarbons.
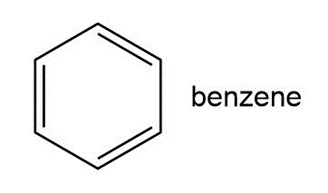
a. How is each carbon atom hybridized?
b. What is the geometry around each carbon atom? What is the overall geometry of the benzene ring?
c. Follow the indicated curved arrow notation to draw a second resonance structure.
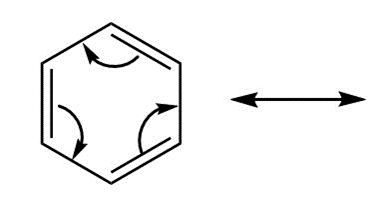
d. Benzene and other aromatic hydrocarbons are shown in Chapter 17 to be very stable. Offer an explanation.
Question: Draw in all hydrogens and lone pairs on the charged carbons in each ion.
a.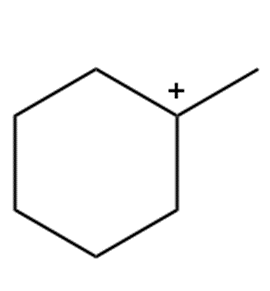
b. 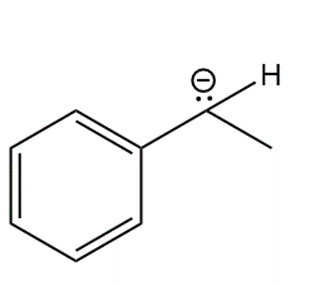
c. 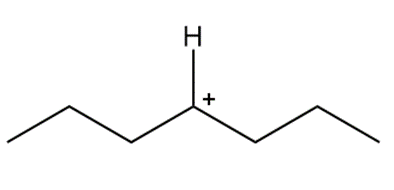
d.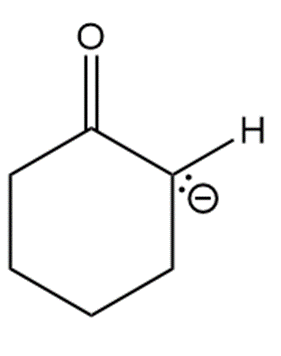
Question: Anacin is an over-the-counter pain reliever that contains aspirin and caffeine. Answer the following questions about each compound:
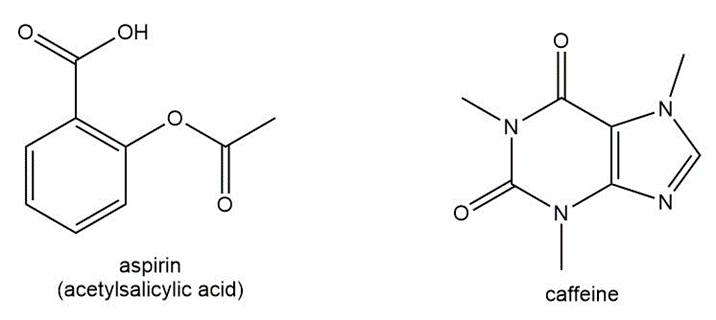
a. What is the molecular formula?
b. How many lone pairs are present on heteroatoms?
c. Label the hybridization state of each carbon.
d. Draw three additional resonance structures.
Question: Draw Lewis structures for the nine isomers having a molecular formula , with all atoms having a zero formal charge.
Question: Use curved arrow notation to show how the first resonance structure can be converted to the second.
a.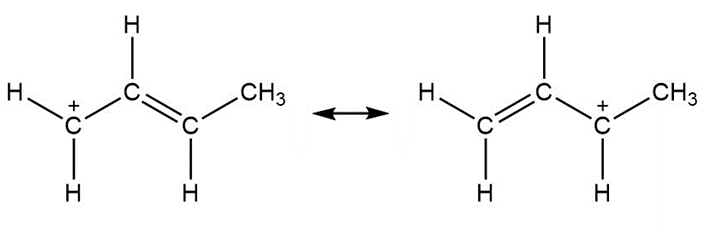
b.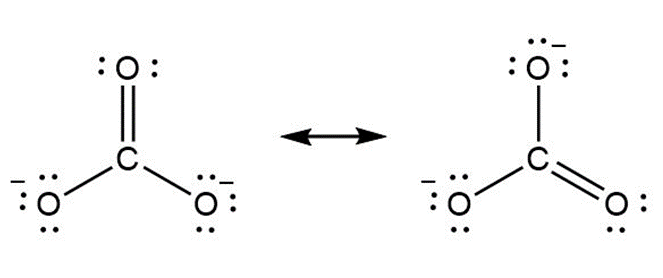
What do you think about this solution?
We value your feedback to improve our textbook solutions.