Chapter 1: Q13. (page 22)
Question: Use curved arrow notation to show how the first resonance structure can be converted to the second.
a.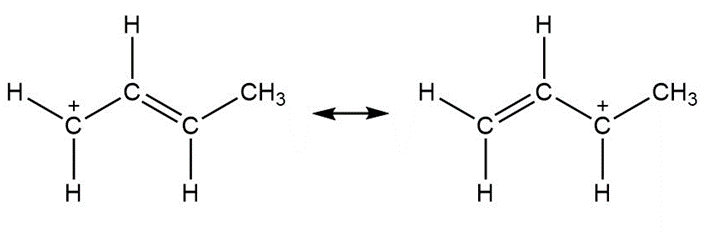
b.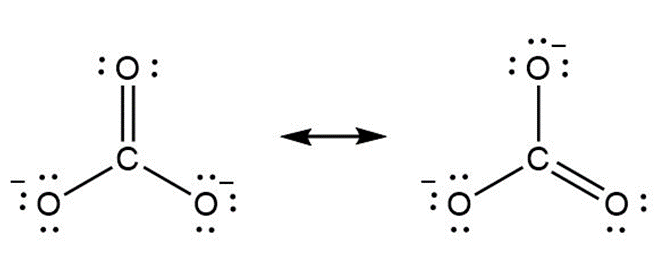
Short Answer
Answer
a.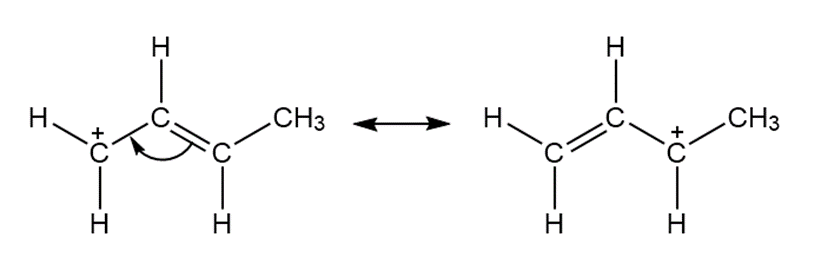
b. 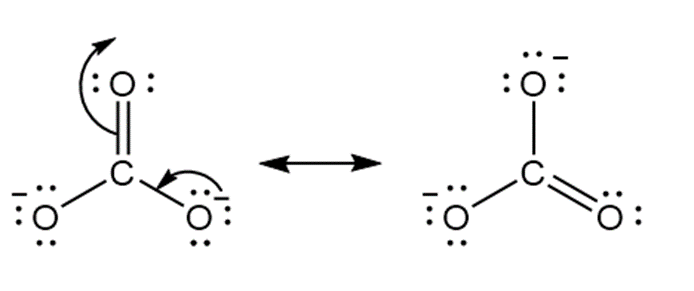
Learning Materials
Features
Discover
Chapter 1: Q13. (page 22)
Question: Use curved arrow notation to show how the first resonance structure can be converted to the second.
a.
b.
Answer
a.
b. 
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Assign formal charges to each carbon atom in the given species. All lone pairs have been drawn in.
a.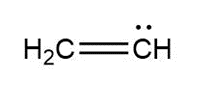
b. 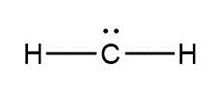
c.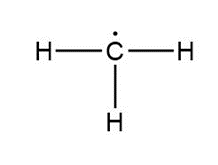
d.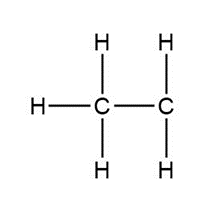
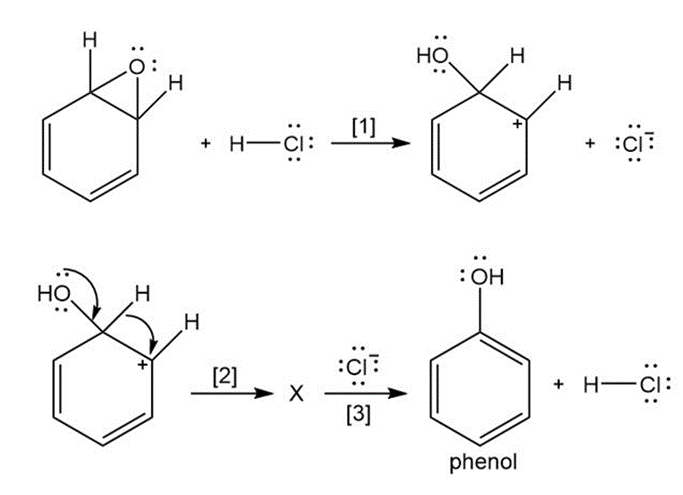
Question: The curved arrow notation introduced in Section 1.6 is a powerful method used by organic chemists to show the movement of electrons not only in resonance structures but also in chemical reactions. Since each curved arrow shows the movement of two electrons, following the curved arrows illustrates what bonds are broken and formed in a reaction. Consider the following three-step process: (a) Add curved arrows in Step [1] to show the movement of electrons. (b) Use the curved arrows drawn in Step [2] to identify the structure of X; X is converted in Step [3] to phenol and HCl.
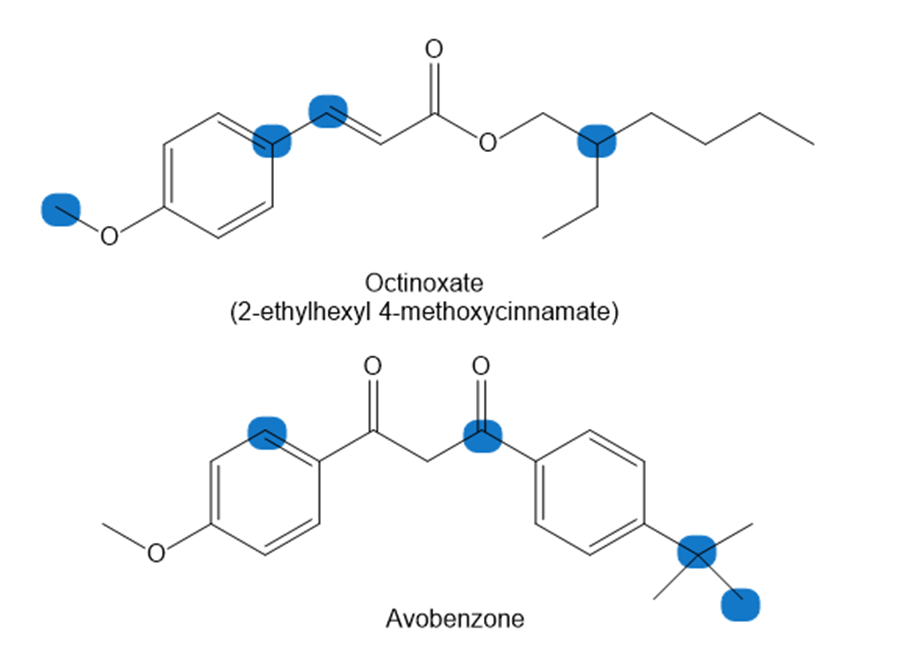
Question: How many hydrogen atoms are present around each highlighted carbon atom in the following molecules? What is the molecular formula for each molecule? Both compounds are active ingredients in some common sunscreens.
Question: Convert each skeletal structure to a complete structure with all C’s, H’s, and lone pairs drawn in.
a.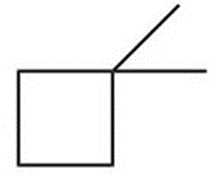
b.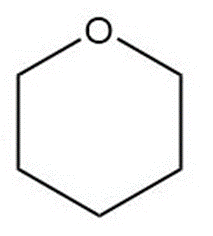
c. 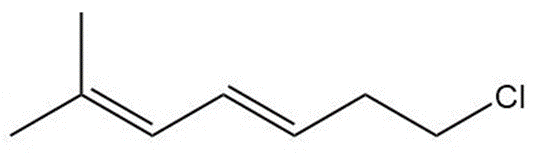
d.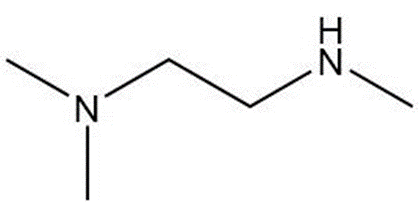
Question: Considering structures, A–D, classify each pair of compounds as isomers, resonance structures, or neither: (a) A and B; (b) A and C; (c) A and D; (d) B and D.
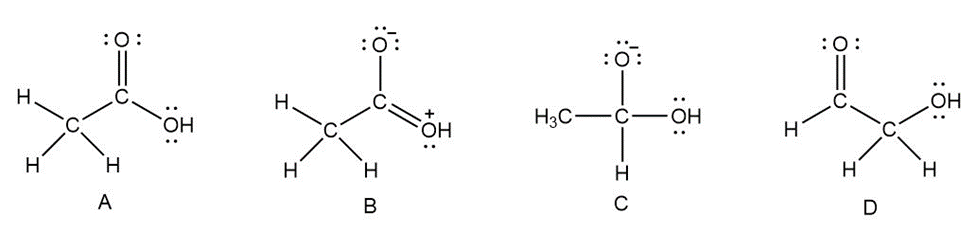
What do you think about this solution?
We value your feedback to improve our textbook solutions.