Chapter 1: Q85. (page 60)
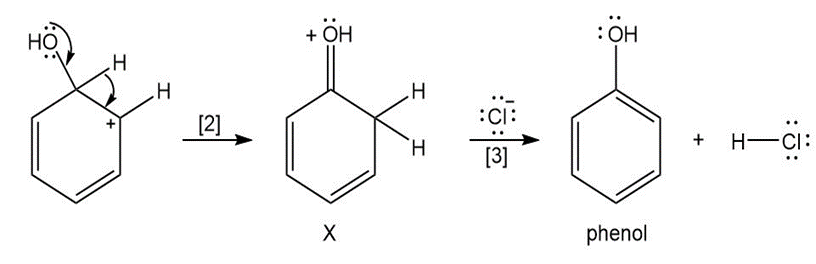
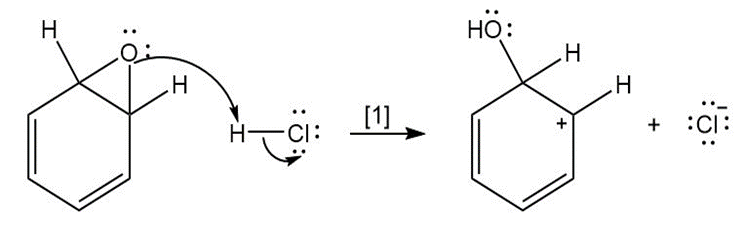
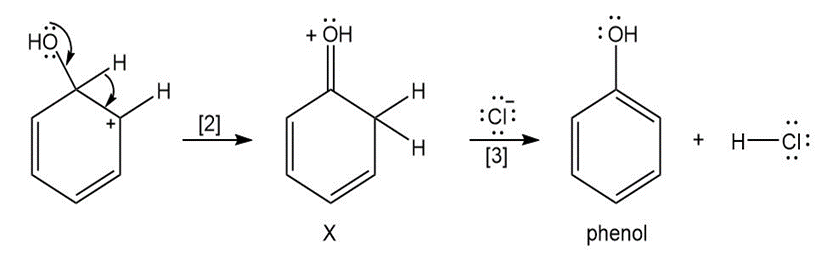
Question: The curved arrow notation introduced in Section 1.6 is a powerful method used by organic chemists to show the movement of electrons not only in resonance structures but also in chemical reactions. Since each curved arrow shows the movement of two electrons, following the curved arrows illustrates what bonds are broken and formed in a reaction. Consider the following three-step process: (a) Add curved arrows in Step [1] to show the movement of electrons. (b) Use the curved arrows drawn in Step [2] to identify the structure of X; X is converted in Step [3] to phenol and HCl.
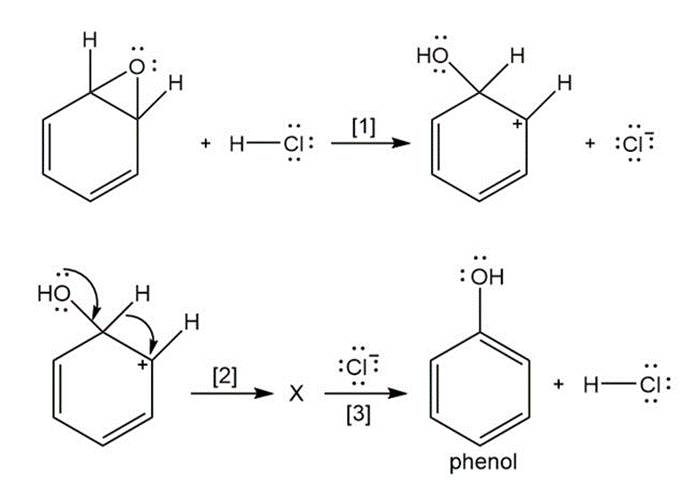
Short Answer
Answer
a.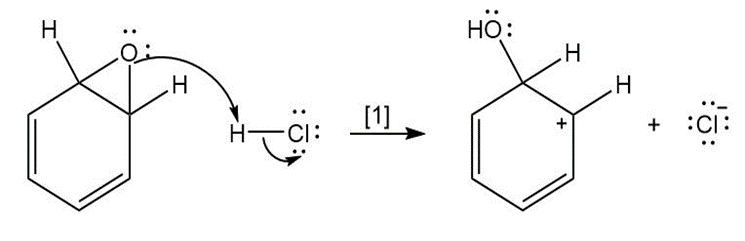
b.