Chapter 1: Q84. (page 60)
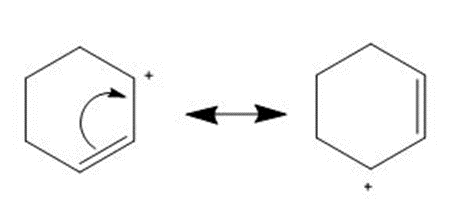
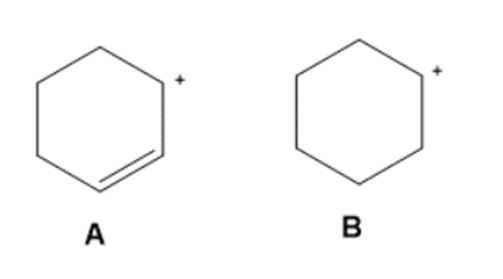
Question: Use the principles of resonance theory to explain why carbocation A is more stable than carbocation B.
Short Answer
Answer
Carbocation A is more stable than carbocation B as the positive charge is distributed over the two carbons because of resonance.