Chapter 1: Q83. (page 59)
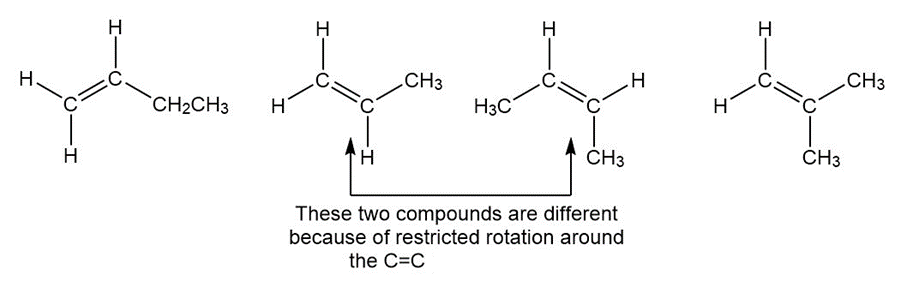
Question: Draw all possible isomers having a molecular formula C4H8 that contains one π bond.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 1: Q83. (page 59)
Question: Draw all possible isomers having a molecular formula C4H8 that contains one π bond.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
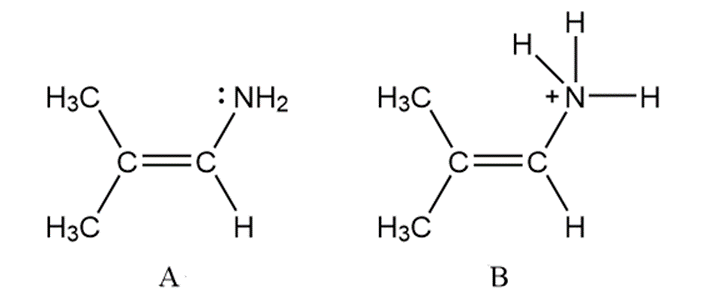
Question: (a) Draw a second resonance structure for A. (b) Why can’t a second resonance
structure be drawn for B?
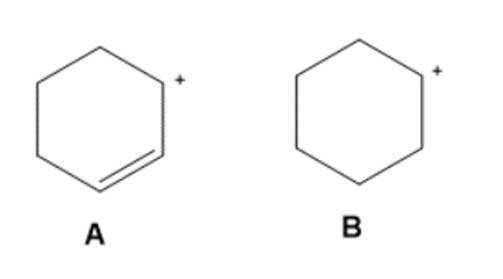
Question: Use the principles of resonance theory to explain why carbocation A is more stable than carbocation B.
Question: Draw a Lewis structure for each ion.
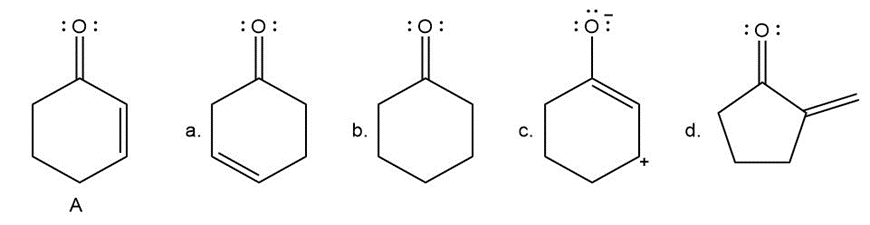
Question: With reference to compound A drawn below, label each compound as an isomer, a resonance structure, or neither.
Question: Draw an acceptable Lewis structure for each compound, assuming the atoms are connected as arranged. Hydrogen cyanideis a poison, formaldehyde is a preservative, and glycolic acid is used to make dissolving sutures.
What do you think about this solution?
We value your feedback to improve our textbook solutions.