Chapter 1: Q20. (page 32)
Question: Convert each condensed formula to a Lewis structure
Short Answer
Answer
a.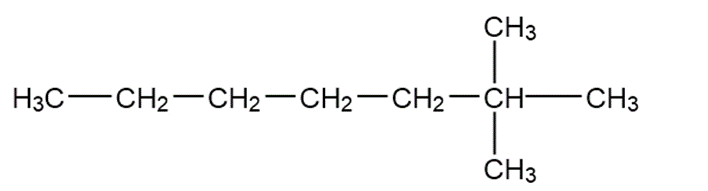
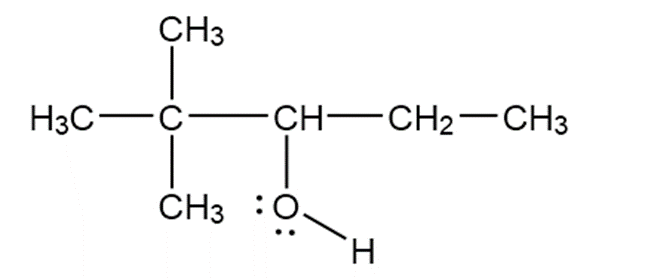
b.
c. 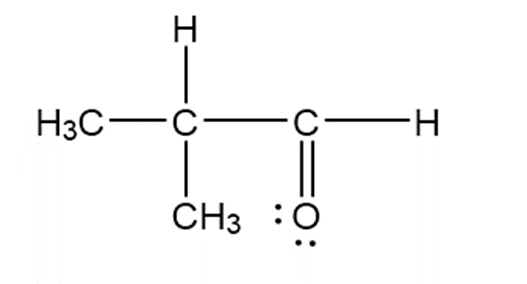
d.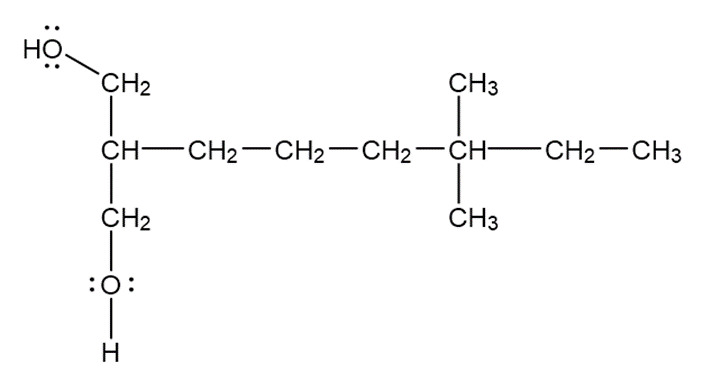
Learning Materials
Features
Discover
Chapter 1: Q20. (page 32)
Question: Convert each condensed formula to a Lewis structure
Answer
a.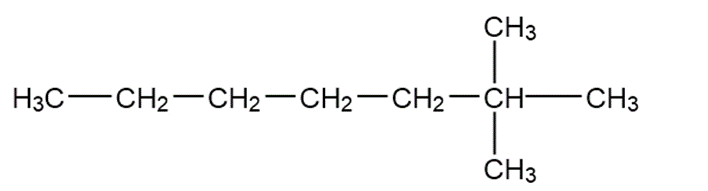

b.
c. 
d.
All the tools & learning materials you need for study success - in one app.
Get started for free
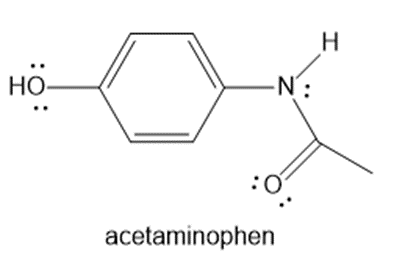
Question: Draw at least ten more resonance structures for acetaminophen, the active pain reliever in Tylenol.
While the most common isotope of nitrogen has a mass number of 14 (nitrogen-14), a radioactive isotope of nitrogen has a mass number of 13 (nitrogen-13). Nitrogen-13 is used in PET (positron emission tomography) scans by physicians to monitor brain activity and diagnose dementia. For each isotope, give the following information: (a) the number of protons; (b) the number of neutrons; (c) the number of electrons in the neutral atom; (d) the group number; and (e) the number of valence electrons.
Question: Determine the hybridization around the highlighted atoms in each molecule.
a. 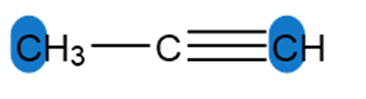
b. 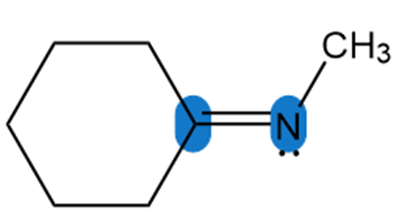
c.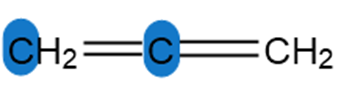
Question: Use the symbols and toindicate the polarity of the highlighted bonds.
a.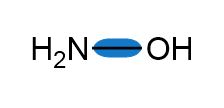
b.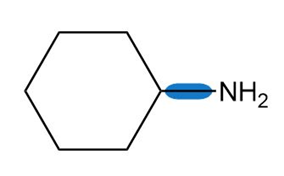
c.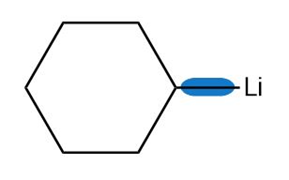
Question: Draw a second resonance structure for each species in parts (a) and (b). Draw two additional resonance structures for the ion in part (c).
a.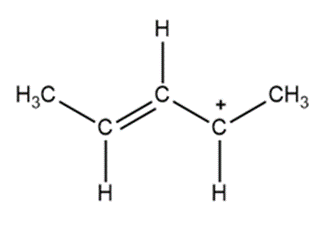
b.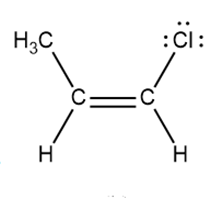
c.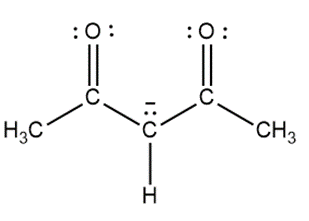
What do you think about this solution?
We value your feedback to improve our textbook solutions.