Chapter 1: Q39. (page 53)
Question: Assign formal charges to each carbon atom in the given species. All lone pairs have been drawn in.
a.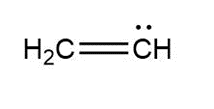
b. 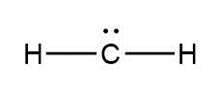
c.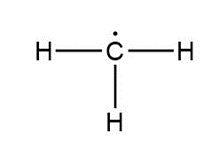
d.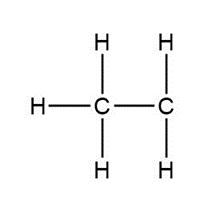
Short Answer
Answer
a. 0 and -1, respectively
b. 0
c. 0
d. 0 and +1, respectively
Learning Materials
Features
Discover
Chapter 1: Q39. (page 53)
Question: Assign formal charges to each carbon atom in the given species. All lone pairs have been drawn in.
a.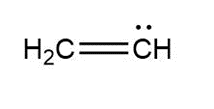
b. 
c.
d.
Answer
a. 0 and -1, respectively
b. 0
c. 0
d. 0 and +1, respectively
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Determine the hybridization around the highlighted atoms in each molecule.
a. 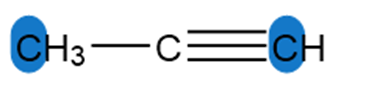
b. 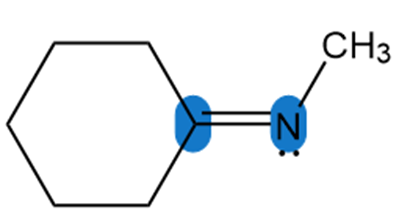
c.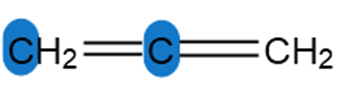
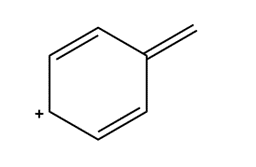
Question: Draw four additional resonance structures for the following cation. Then draw the resonance hybrid.
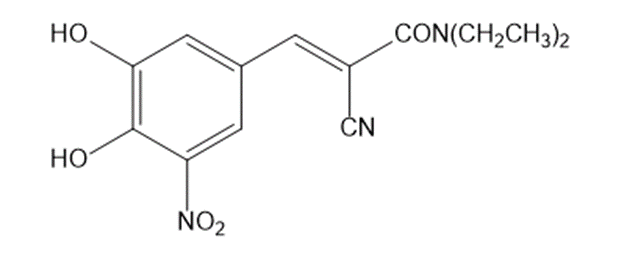
Question: Stalevo is the trade name for a medication used in Parkinson’s disease, containing L-dopa, carbidopa, and entacapone.
a. Draw a Lewis structure for entacapone.
b. Which C–C bond in entacapone is the longest?
c. Which C–C single bond is the shortest?
d. Which C–N bond is the longest?
e. Which C–N bond is the shortest?
f. Use curved arrows to draw a resonance structure that is an equal contributor to the resonance hybrid.
g. Use curved arrows to draw a resonance structure that is a minor contributor to the resonance hybrid.
Question: Show the direction of the dipole in each bond. Label the atoms with .
a. 
b. 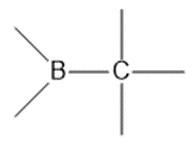
c. 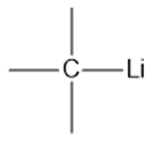
d. 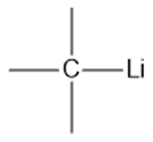
What do you think about this solution?
We value your feedback to improve our textbook solutions.