Chapter 1: Q6. (page 17)
Question: Calculate the formal charge on each second-row atom
a. 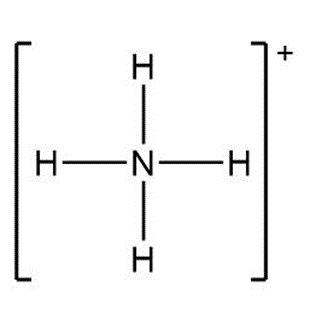
b. 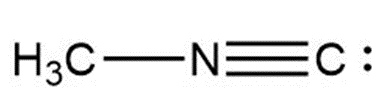
c.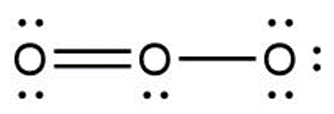
Short Answer
Answer
a. +1
b. 0, +1, -1 (from left to right)
c. 0, +1, -1 (from left to right)
Learning Materials
Features
Discover
Chapter 1: Q6. (page 17)
Question: Calculate the formal charge on each second-row atom
a. 
b. 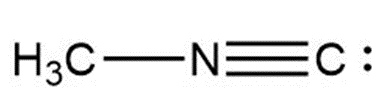
c.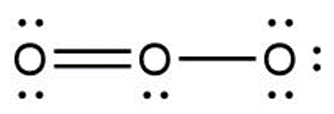
Answer
a. +1
b. 0, +1, -1 (from left to right)
c. 0, +1, -1 (from left to right)
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Zingerone gives ginger its pungent taste.
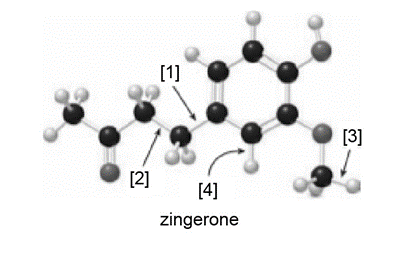
a. What is the molecular formula for zingerone?
b. How many lone pairs are present?
c. Draw a skeletal structure.
d. How many hybridized carbons are present?
e. What orbitals are used to form each indicated bond ([1]–[4])?
Question: Ketene, , is an unusual organic molecule that has a single carbon atom doubly bonded to two different atoms. Determine the hybridization of both C and the O in ketene. Then, draw a diagram showing what orbitals are used to form each bond (similar to Figures 1.10 and 1.11).
Question: Draw in all the carbon and hydrogen atoms in each molecule.
a.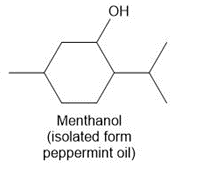
b.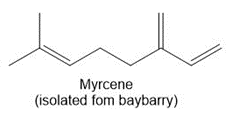
c.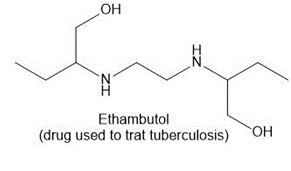
d.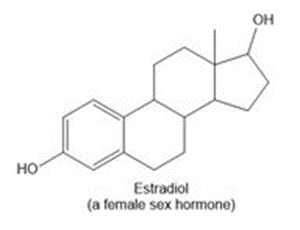
Question: Convert each molecule into a skeletal structure.
a.
b.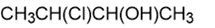
c.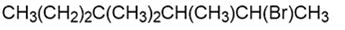
d.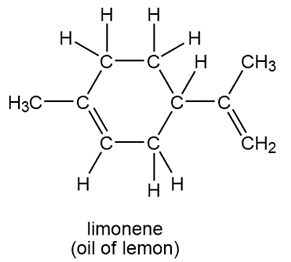
Question: Label each bond in the following compounds as ionic or covalent.
What do you think about this solution?
We value your feedback to improve our textbook solutions.