Chapter 1: Q54. (page 55)
Question: Draw four additional resonance structures for the following cation. Then draw the resonance hybrid.
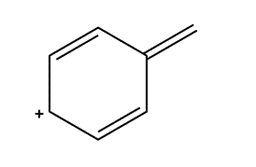
Short Answer
Answer
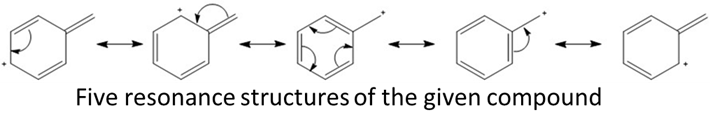
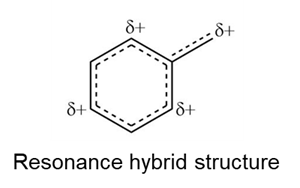
Learning Materials
Features
Discover
Chapter 1: Q54. (page 55)
Question: Draw four additional resonance structures for the following cation. Then draw the resonance hybrid.

Answer


All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Consider compounds A–D, which contain both a heteroatom and a double bond. (a) For which of the compounds are no additional Lewis structures possible? (b) When two or more Lewis structures can be drawn, draw all additional resonance structures.
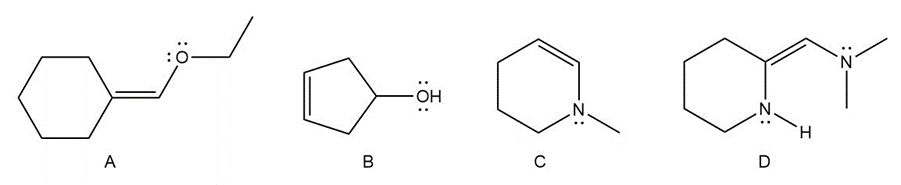
Question: How many covalent bonds are predicted for each atom?
Question: Benzene is the simplest member of a whole class of compounds called aromatic hydrocarbons.
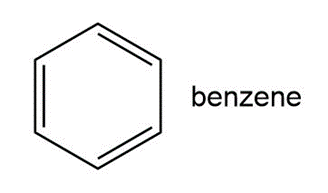
a. How is each carbon atom hybridized?
b. What is the geometry around each carbon atom? What is the overall geometry of the benzene ring?
c. Follow the indicated curved arrow notation to draw a second resonance structure.
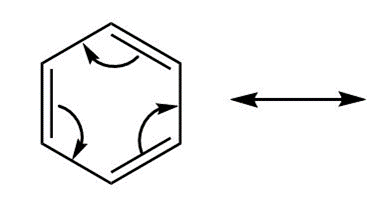
d. Benzene and other aromatic hydrocarbons are shown in Chapter 17 to be very stable. Offer an explanation.
Question: Draw in all the hydrogen atoms and nonbonded electron pairs in each ion.
a.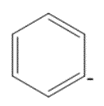
b.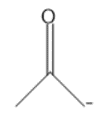
c.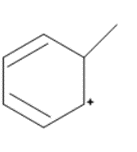
d.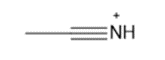
e.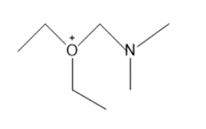
Question: Label the polar bonds in each molecule. Indicate the direction of the net dipole (if there is one).
c.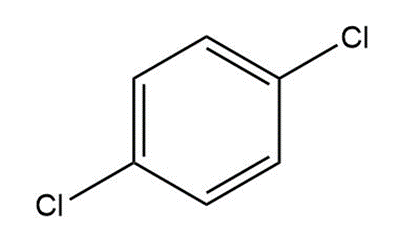
d.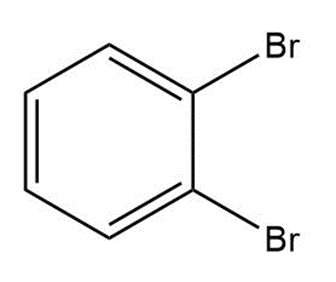
What do you think about this solution?
We value your feedback to improve our textbook solutions.