Chapter 1: Q12. (page 22)
Question: Follow the curved arrows to draw a second resonance structure for each species.
a. 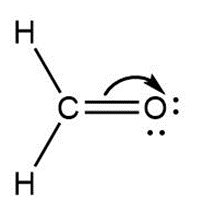
b.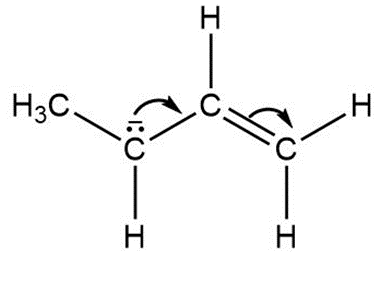
Short Answer
Answer
a.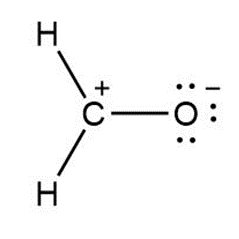
b. 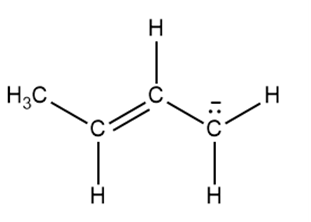
Learning Materials
Features
Discover
Chapter 1: Q12. (page 22)
Question: Follow the curved arrows to draw a second resonance structure for each species.
a. 
b.
Answer
a.
b. 
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Indicate which of the following molecules is polar because it possesses a net dipole. Show the direction of the net dipole if one exists.
d.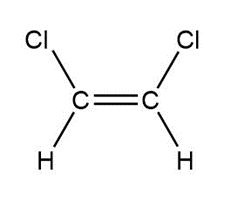
e.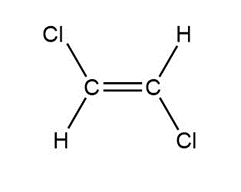
Question: Answer the following questions about compound A:
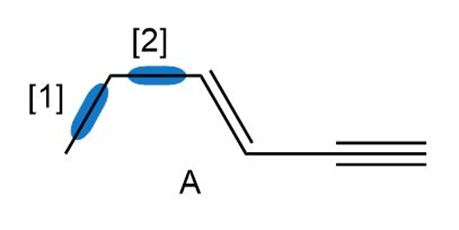
Question: How are the molecules or ions in each pair related? Classify them as resonance structures, isomers, or neither.
a. 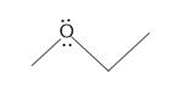
and
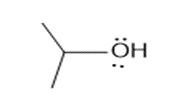
b. and
and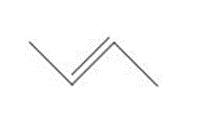
c.  and
and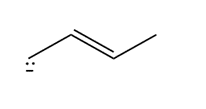
d. and
and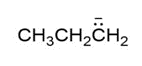
Question: Draw Lewis structures for each molecular formula.
a. C2H4Cl2( two isomers)
b. C3H8O (three isomers)
c. C3H6 (two isomers)
Question: Predict the hybridization and geometry around each highlighted atom.
a.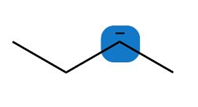
b.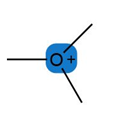
c.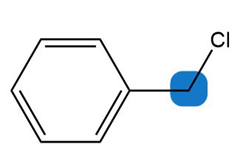
d.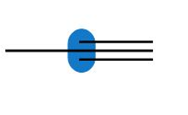
e.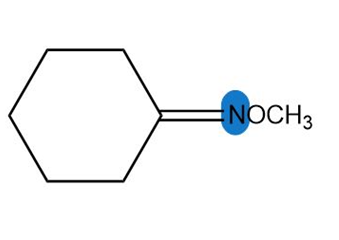
What do you think about this solution?
We value your feedback to improve our textbook solutions.