Chapter 1: Q45. (page 54)
Question: Draw Lewis structures for the nine isomers having a molecular formula , with all atoms having a zero formal charge.
Short Answer
Answer
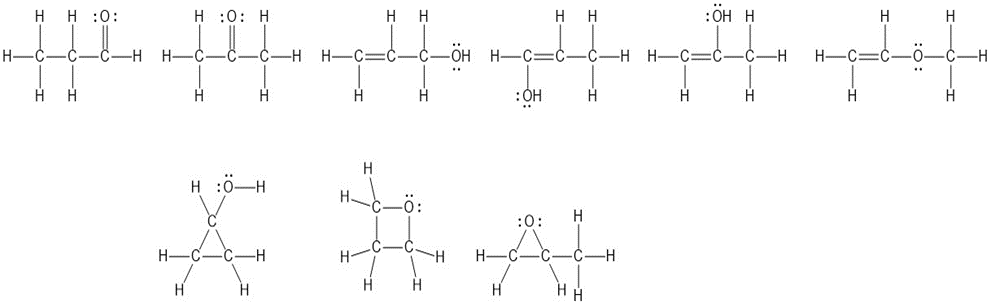
The Lewis structures of the nine isomers are given below:
Learning Materials
Features
Discover
Chapter 1: Q45. (page 54)
Question: Draw Lewis structures for the nine isomers having a molecular formula , with all atoms having a zero formal charge.
Answer
The Lewis structures of the nine isomers are given below:

All the tools & learning materials you need for study success - in one app.
Get started for free
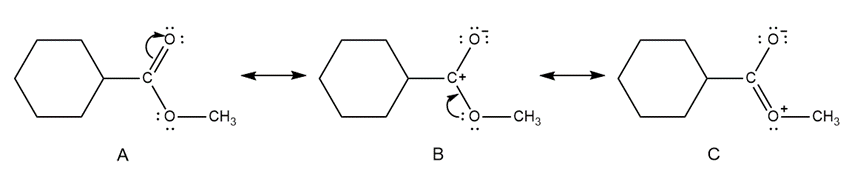
Question: Which of the given resonance structures (A, B, or C) contributes most to the resonance hybrid? Which contributes the least?
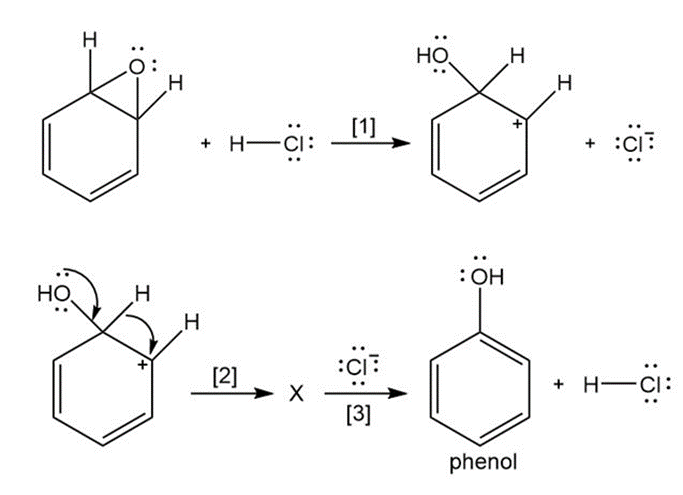
Question: The curved arrow notation introduced in Section 1.6 is a powerful method used by organic chemists to show the movement of electrons not only in resonance structures but also in chemical reactions. Since each curved arrow shows the movement of two electrons, following the curved arrows illustrates what bonds are broken and formed in a reaction. Consider the following three-step process: (a) Add curved arrows in Step [1] to show the movement of electrons. (b) Use the curved arrows drawn in Step [2] to identify the structure of X; X is converted in Step [3] to phenol and HCl.
Question: Convert each condensed formula to a Lewis structure
Question: Two useful organic compounds that contain Cl atoms are vinyl chloride and chloroethane . Vinyl chloride is the starting material used to prepare poly (vinyl chloride), a plastic used in insulation, pipes, and bottles. Chloroethane (ethyl chloride) is a local anesthetic. Why is the bond of vinyl chloride stronger than the bond in chloroethane?
Question: Zingerone gives ginger its pungent taste.
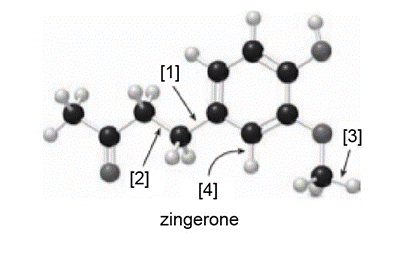
a. What is the molecular formula for zingerone?
b. How many lone pairs are present?
c. Draw a skeletal structure.
d. How many hybridized carbons are present?
e. What orbitals are used to form each indicated bond ([1]–[4])?
What do you think about this solution?
We value your feedback to improve our textbook solutions.