Chapter 1: Q18. (page 29)
Question: Predict the indicated bond angles in each compound drawn as a Lewis structure with no implied geometry.
a. 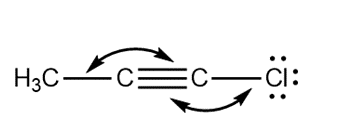
b. 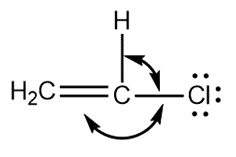
c.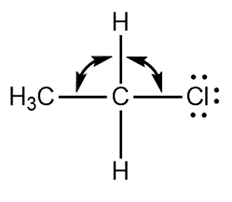
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 1: Q18. (page 29)
Question: Predict the indicated bond angles in each compound drawn as a Lewis structure with no implied geometry.
a. 
b. 
c.
Answer
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Citric acid is responsible for the tartness of citrus fruits, especially lemons and limes.
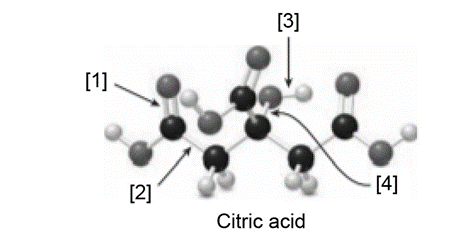
a. What is the molecular formula for citric acid?
b. How many lone pairs are present?
c. Draw a skeletal structure.
d. How many hybridized carbons are present?
e. What orbitals are used to form each indicated bond ([1]–[4])?
Question: What orbitals are used to form each of the C-C and C-H bonds in (propane)? How many bonds are present in this molecule?
Question: Draw Lewis structures for the nine isomers having a molecular formula , with all atoms having a zero formal charge.
Question: Draw a skeletal structure for the molecules in parts (a) and (b), and a condensed structure for the molecules in parts (c) and (d).
a. 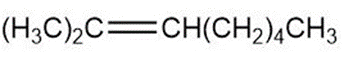
b. 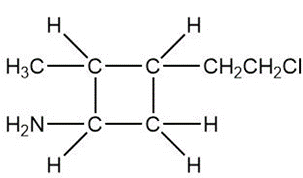
c. 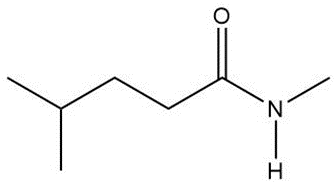
d.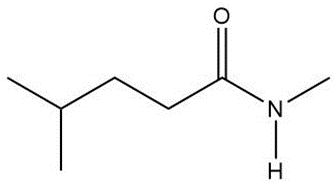
Question: Draw in all the carbon and hydrogen atoms in each molecule.
a.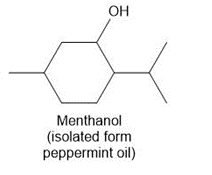
b.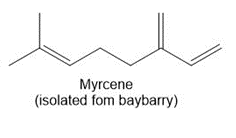
c.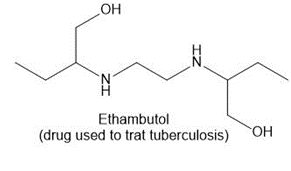
d.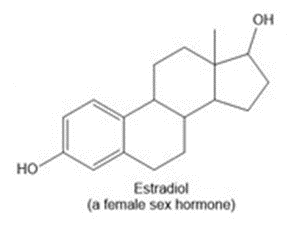
What do you think about this solution?
We value your feedback to improve our textbook solutions.