Amygdalin, a compound isolated from the pits of apricots, peaches, and wild cherries, has been used as an unsanctioned anticancer drug both within and outside of the United States. One hydrolysis product formed from amygdalin is mandelic acid, used in treating common skin problems caused by photo-aging and acne.
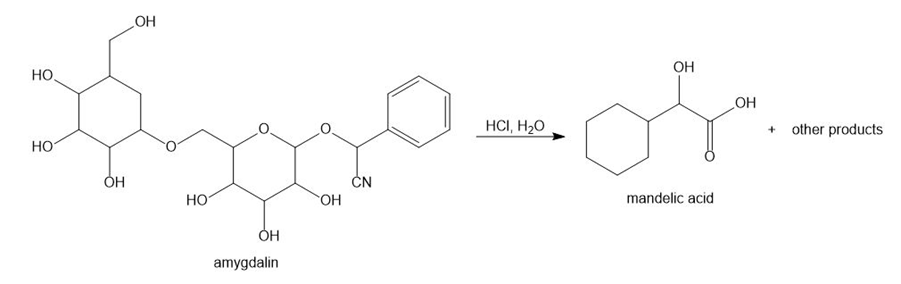
a. How many stereogenic centres are present in amygdalin? What is the maximum number of stereoisomers possible?
b. Draw both enantiomers of mandelic acid and label each stereogenic centre as R or S.
c. Pure (R)-mandelic acid has a specific rotation of –154. If a sample contains 60% of the R isomer and 40% of its enantiomer, what is of this solution?
d. Calculate the ee of a solution of mandelic acid having = +50. What is the percentage of each enantiomer present?


 Enantiomers of amphetamine
Enantiomers of amphetamine Enantiomers of ketoprofen
Enantiomers of ketoprofen Enantiomers of amphetamine
Enantiomers of amphetamine Enantiomers of ketoprofen
Enantiomers of ketoprofen






