Chapter 5: Q.20 (page 194)
Which compounds are meso compounds?
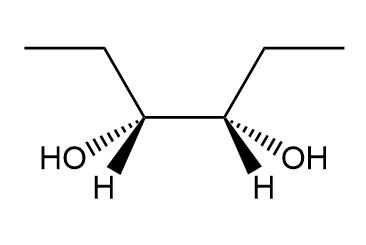
a.
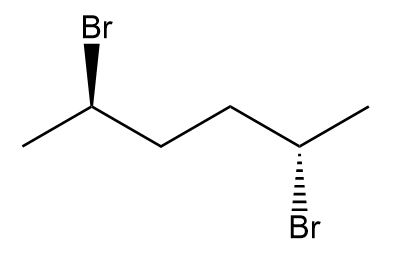
b.
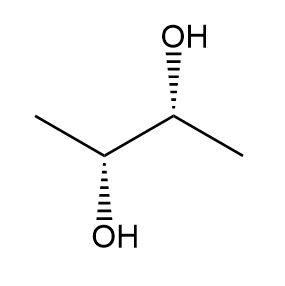
c.
Short Answer
a. Meso compound.
b. Not a meso compound.
c. Meso compound.
Learning Materials
Features
Discover
Chapter 5: Q.20 (page 194)
Which compounds are meso compounds?

a.

b.

c.
a. Meso compound.
b. Not a meso compound.
c. Meso compound.
All the tools & learning materials you need for study success - in one app.
Get started for free
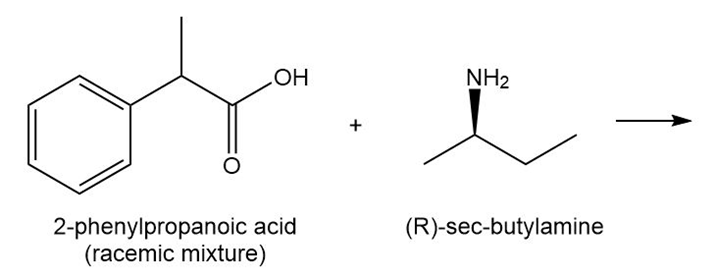
An acid–base reaction of (R)-sec-butylamine with a racemic mixture of 2-phenylpropanoic acid forms two products having different melting points and somewhat different solubilities. Draw the structure of these two products. Assign R and S to any stereogenic centers in the products. How are the two products related? Choose from enantiomers, diastereomers, constitutional isomers, or not isomers.
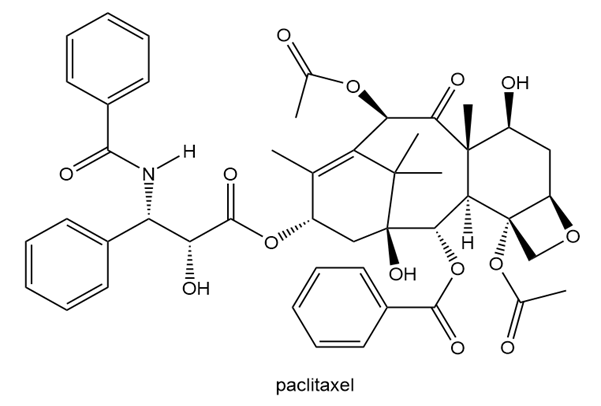
Label the stereogenic centers in paclitaxel, the chapter-opening anticancer drug, as R or S.
A natural product was isolated in the laboratory, and its observed rotation was +10° when measured in a 1 dm sample tube containing 1.0 g of compound in 10 mL of water. What is the specific rotation of this compound?
Draw all possible stereoisomers for each compound. Label pairs of enantiomers and diastereomers. Label any meso compound.
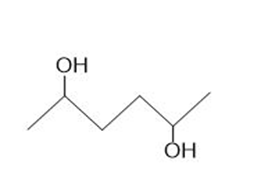
a.
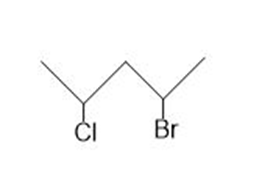
b.
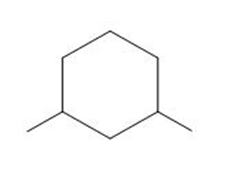
c.
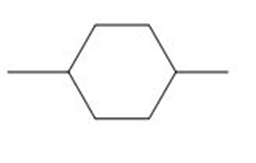
d.
The shrub ma huang (Section 5.4 A) contains two biologically active stereoisomers-ephedrine and pseudoephedrine-with two stereogenic centers as shown in the given structure. Ephedrine is one component of a once popular combination drug used by body builders to increase energy and alertness, while pseudoephedrine is a nasal decongestant.
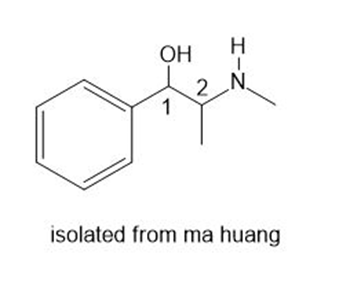
a. Draw the structure of naturally occurring (–)-ephedrine, which has the 1R,2Sconfiguration.
b. Draw the structure of naturally occurring (+)-pseudoephedrine, which has the 1S,2Sconfiguration.
c. How are ephedrine and pseudoephedrine related?
d. Draw all other stereoisomers of (–)-ephedrine and (+)-pseudoephedrine and give the R,Sdesignation for all stereogenic centers.
e. How is each compound drawn in part (d) related to (–)-ephedrine?
What do you think about this solution?
We value your feedback to improve our textbook solutions.