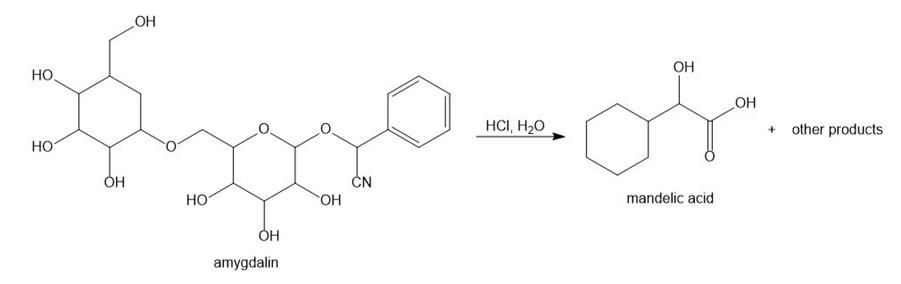
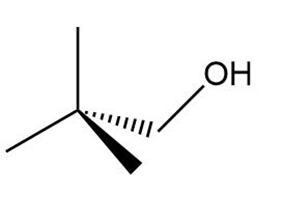
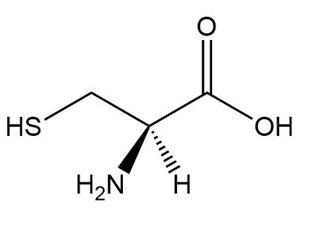
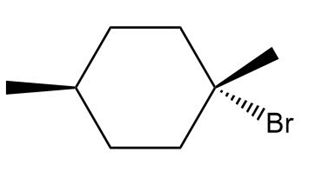
Chapter 5: Q,56 (page 209)
Draw all possible stereoisomers for each compound. Label pairs of enantiomers and diastereomers. Label any meso compound.
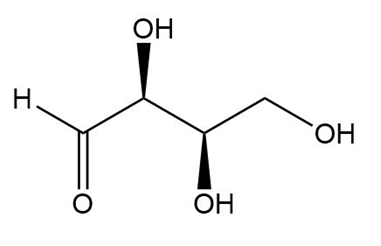
a.
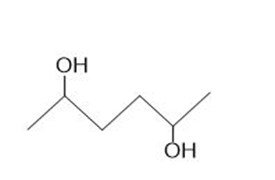
b.
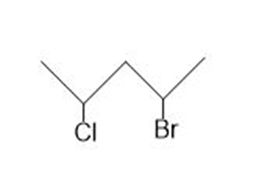
c.
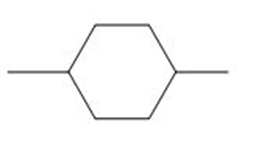
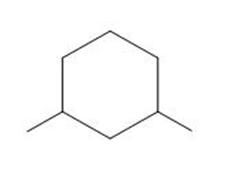
d.
Short Answer
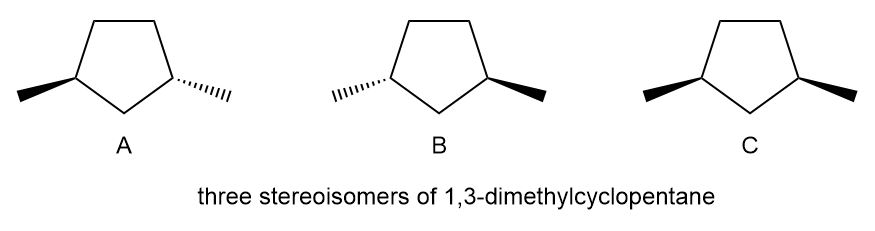
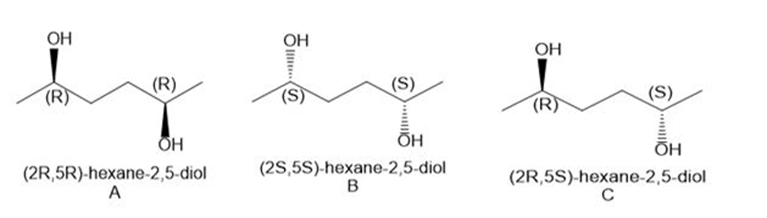
a. The stereoisomers of compound a are given below:
The meso compound of compound a is given below:
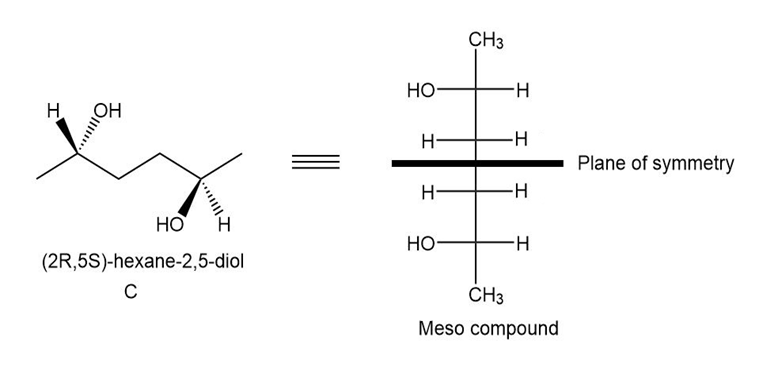
Pair A and B are enantiomers, and pairs A, C, and B, C is diastereomers.
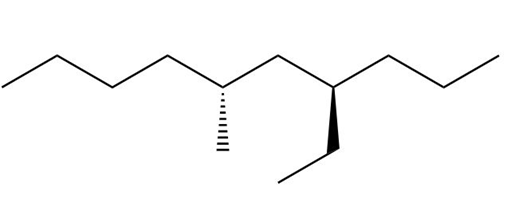
b. The stereoisomers of compound b are given below:
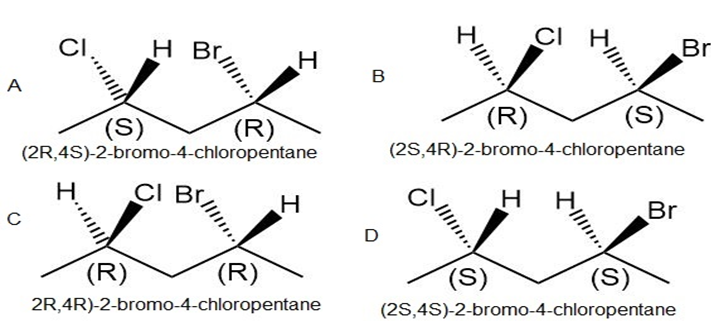
This compound does not contain a meso compound.
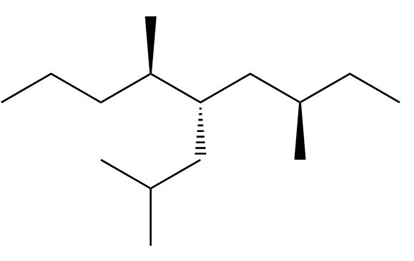
c. The stereoisomers of compound c are given below:
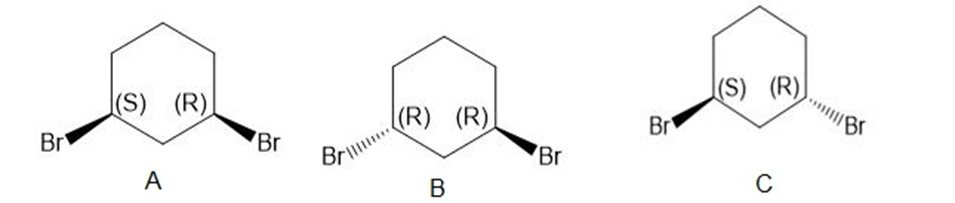
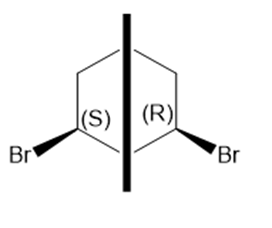
The meso compound of compound c is given below:
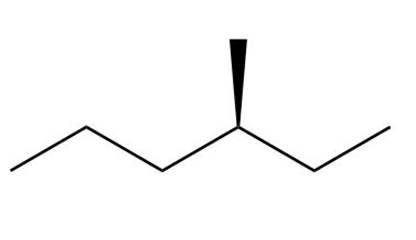
d. This compound does not contain any stereoisomers.
 Stereocenters in compound a
Stereocenters in compound a Stereoisomers in compound a
Stereoisomers in compound a Meso compound of compound a
Meso compound of compound a Stereocenters of the compound b
Stereocenters of the compound b Stereoisomers of the compound b
Stereoisomers of the compound b Stereocenter in compound c
Stereocenter in compound c Stereoisomers of the compound c
Stereoisomers of the compound c Meso compound of the compound c
Meso compound of the compound c