Chapter 5: Q.53 (page 208)
Label the stereogenic centers in paclitaxel, the chapter-opening anticancer drug, as R or S.
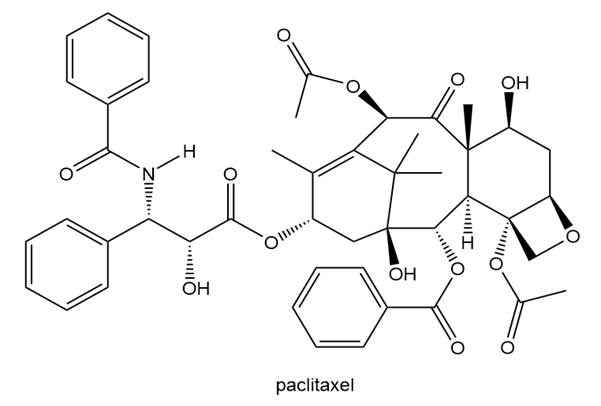
Short Answer
The stereogenic centers of paclitaxel are labeled as follows:
Learning Materials
Features
Discover
Chapter 5: Q.53 (page 208)
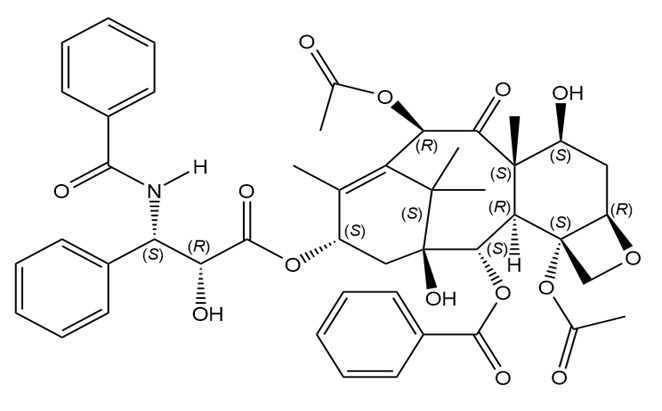
Label the stereogenic centers in paclitaxel, the chapter-opening anticancer drug, as R or S.

The stereogenic centers of paclitaxel are labeled as follows:

All the tools & learning materials you need for study success - in one app.
Get started for free
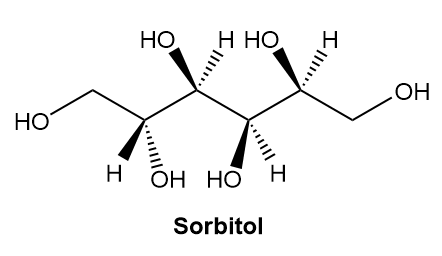
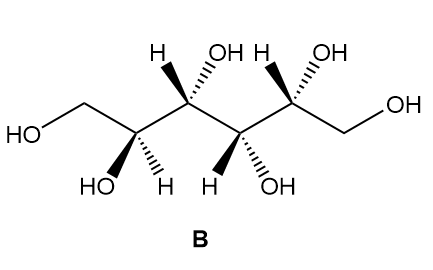
a. Label the four stereogenic centres in sorbitol as R or S.
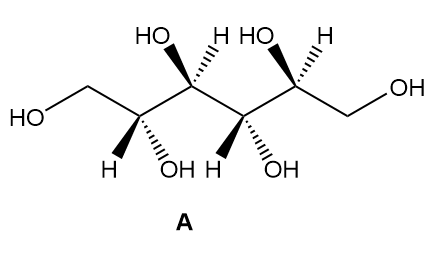
b. How are sorbitol and A related?
c. How are sorbitol and B related?
Label each compound as chiral or achiral. Compounds that contain a single carbon common to two rings are called spiro compounds. Because carbon is tetrahedral, the two rings are perpendicular to each other.
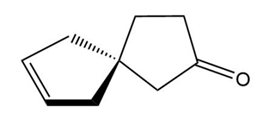
a.
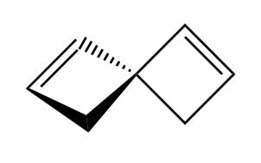
b.
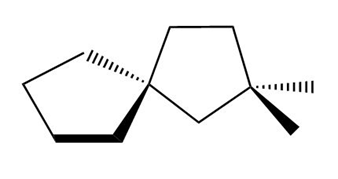
c.
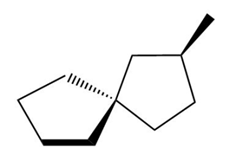
d.
A molecule is achiral if it has a plane of symmetry in any conformation. Each of the following conformations does not have a plane of symmetry, but rotation around a carbon–carbon bond forms a conformation that does have a plane of symmetry. Draw this conformation for each molecule.
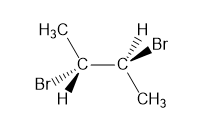
a.
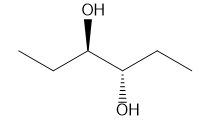
b.
Rank the following groups in order of decreasing priority?
a.
b.
c.
d.
How is each compound related to the simple sugar D-erythrose? Is it an enantiomer,diastereomer, or identical?
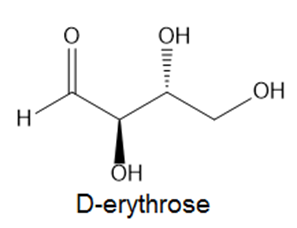
a.
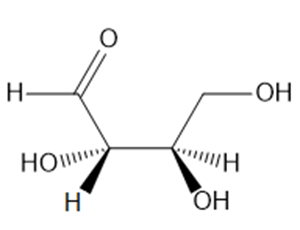
b.
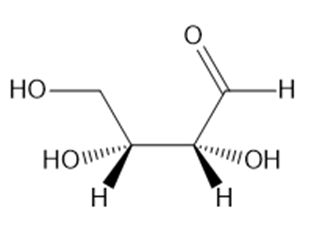
c.
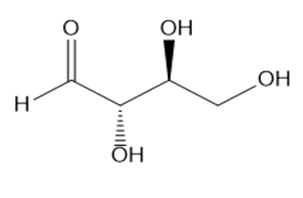
What do you think about this solution?
We value your feedback to improve our textbook solutions.