Chapter 5: Q.24 (page 195)
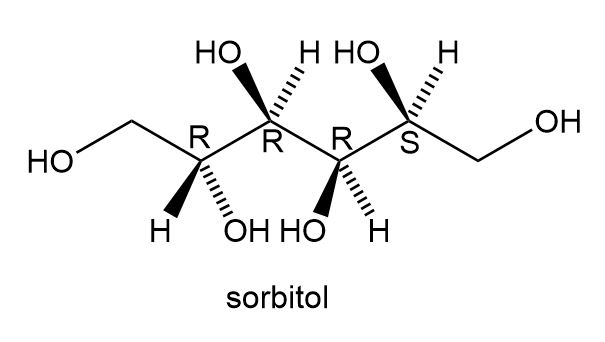
a. Label the four stereogenic centres in sorbitol as R or S.
b. How are sorbitol and A related?
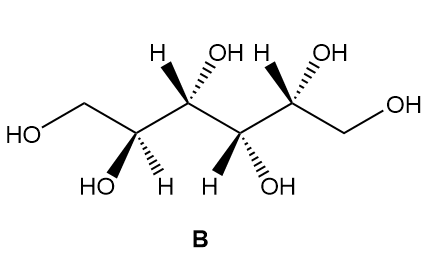
c. How are sorbitol and B related?
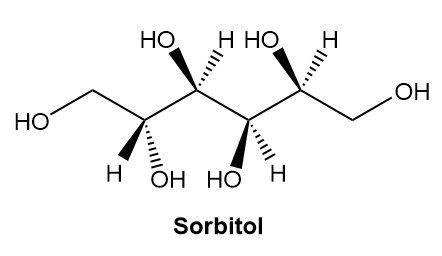

Short Answer
a.

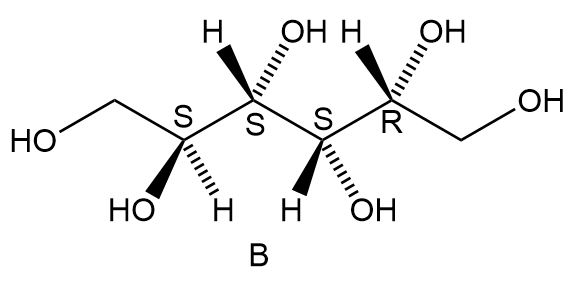
b. Diastereomers.
c. Enantiomers.
Learning Materials
Features
Discover
Chapter 5: Q.24 (page 195)
a. Label the four stereogenic centres in sorbitol as R or S.
b. How are sorbitol and A related?
c. How are sorbitol and B related?



a.



b. Diastereomers.
c. Enantiomers.
All the tools & learning materials you need for study success - in one app.
Get started for free
A molecule is achiral if it has a plane of symmetry in any conformation. Each of the following conformations does not have a plane of symmetry, but rotation around a carbon–carbon bond forms a conformation that does have a plane of symmetry. Draw this conformation for each molecule.
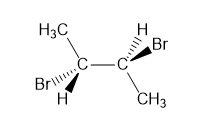
a.
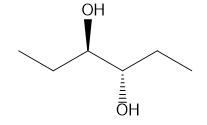
b.
The amino acid (S)-alanine has the physical characteristics listed under the structure.

a. What is the melting point of (R)-alanine?
b. How does the melting point of a racemic mixture of (R)- and (S)-alanine compare to the melting point of (S)-alanine?
c. What is the specific rotation of (R)-alanine, recorded under the same conditions as the reported rotation of (S)-alanine?
d. What is the optical rotation of a racemic mixture of (R)- and (S)-alanine?
e. Label each of the following as optically active or inactive: a solution of pure (S)-alanine; an equal mixture of (R)- and (S)-alanine; a solution that contains 75% (S)- and 25% (R)-alanine.
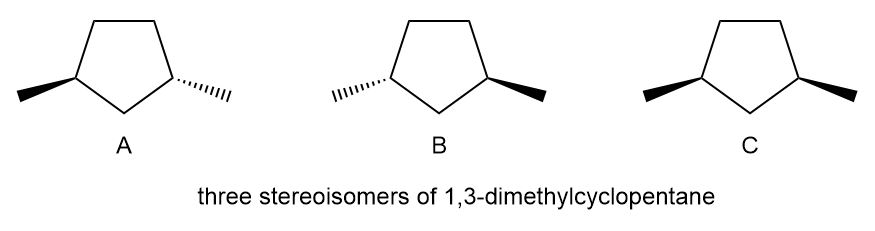
Compare the physical properties of the three stereoisomers of 1,3-dimethylcyclopenatane.
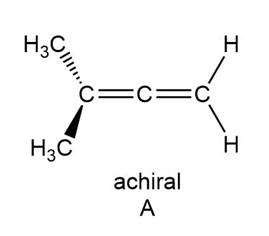
A limited number of chiral compounds having no stereogenic centers exist. For example, although A is achiral, constitutional isomer B is chiral. Make models and explain this observation. Compounds containing two double bonds that share a single carbon atom are called allenes. Locate the allene in the antibiotic mycomycin and decide whether mycomycin is chiral or achiral.
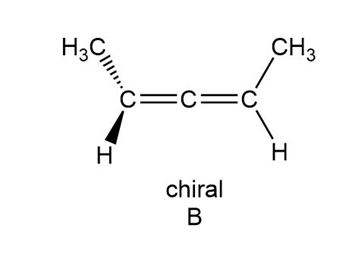
How are the compounds in each pair related to each other? Are they identical, enantiomers, diastereomers, constitutional isomers, or not isomers of each other?
a.
 and
and 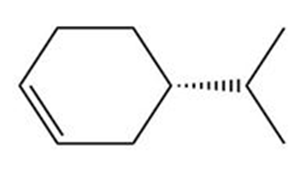
b.
 and
and 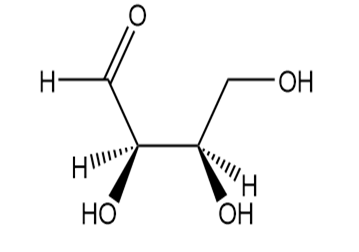
c.
 and
and
d.
 and
and 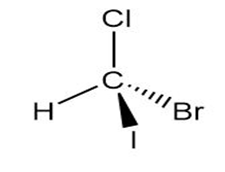
e.
 and
and 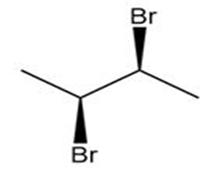
f.
 and
and 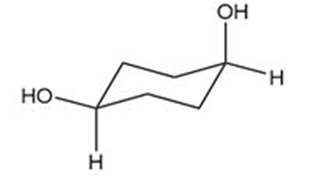
What do you think about this solution?
We value your feedback to improve our textbook solutions.