Chapter 5: Q.45 (page 207)
Which group in each pair is assigned the higher priority in R,S nomenclature?
- localid="1648446360910"
- localid="1648447310055"
Short Answer
The following group among the pairs will get the higher priority:
Learning Materials
Features
Discover
Chapter 5: Q.45 (page 207)
Which group in each pair is assigned the higher priority in R,S nomenclature?
The following group among the pairs will get the higher priority:
All the tools & learning materials you need for study success - in one app.
Get started for free
Determine if each compound is identical to or an enantiomer of A.
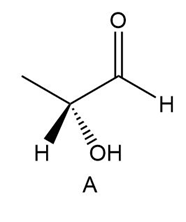
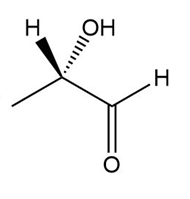
a.
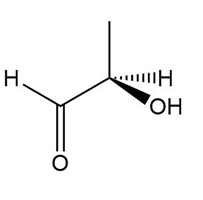
b.
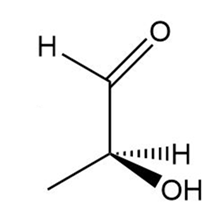
c.
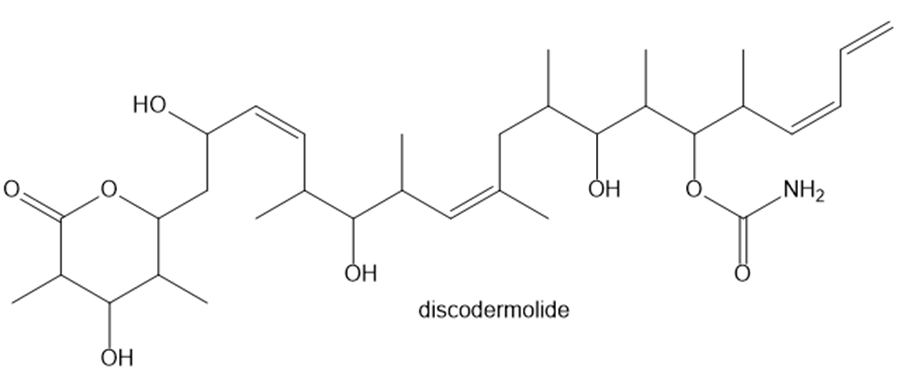
a. Locate all the tetrahedral stereogenic centers in discodermolide, a tumor inhibitor isolated from the Caribbean marine sponge Discodermiadissoluta.
b. Certain carbon–carbon double bonds can also be stereogenic centers. With reference to the definition in Section 5.3, explain how this can occur, and then locate the three additional stereogenic centers in discodermolide.
c. Considering all stereogenic centers, what is the maximum number of stereoisomers possible for discodermolide?
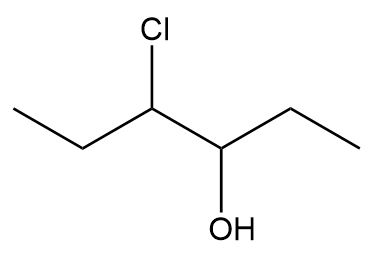
Label the two stereogenic centers in each compound and draw all possible stereoisomers.
a.
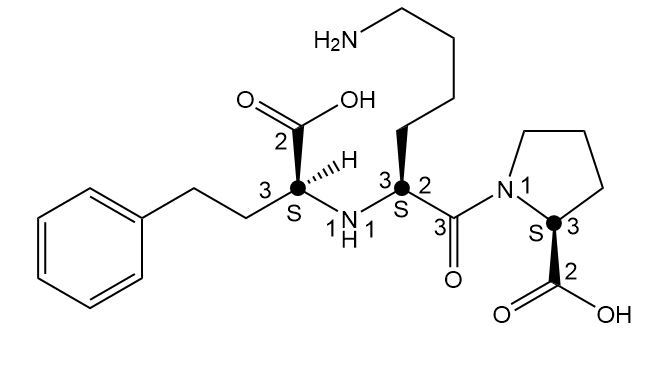
b.
The amino acid (S)-alanine has the physical characteristics listed under the structure.
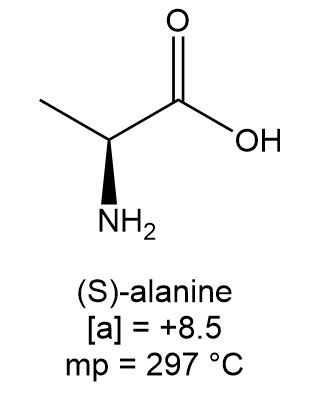
a. What is the melting point of (R)-alanine?
b. How does the melting point of a racemic mixture of (R)- and (S)-alanine compare to the melting point of (S)-alanine?
c. What is the specific rotation of (R)-alanine, recorded under the same conditions as the reported rotation of (S)-alanine?
d. What is the optical rotation of a racemic mixture of (R)- and (S)-alanine?
e. Label each of the following as optically active or inactive: a solution of pure (S)-alanine; an equal mixture of (R)- and (S)-alanine; a solution that contains 75% (S)- and 25% (R)-alanine.
Draw the mirror image of each compound. Label each molecule as chiral or achiral.
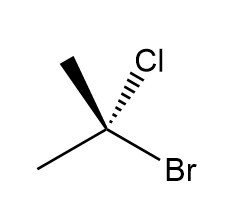
a.
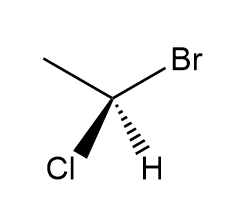
b.
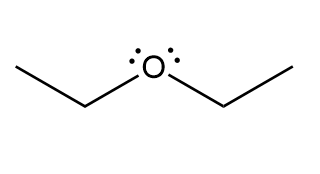
c.
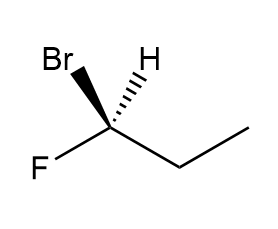
d.
What do you think about this solution?
We value your feedback to improve our textbook solutions.