The shrub ma huang (Section 5.4 A) contains two biologically active stereoisomers-ephedrine and pseudoephedrine-with two stereogenic centers as shown in the given structure. Ephedrine is one component of a once popular combination drug used by body builders to increase energy and alertness, while pseudoephedrine is a nasal decongestant.
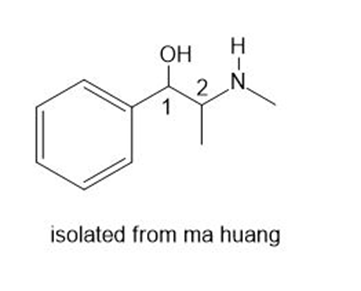
a. Draw the structure of naturally occurring (–)-ephedrine, which has the 1R,2Sconfiguration.
b. Draw the structure of naturally occurring (+)-pseudoephedrine, which has the 1S,2Sconfiguration.
c. How are ephedrine and pseudoephedrine related?
d. Draw all other stereoisomers of (–)-ephedrine and (+)-pseudoephedrine and give the R,Sdesignation for all stereogenic centers.
e. How is each compound drawn in part (d) related to (–)-ephedrine?


 Stereogenic centers in aliskren
Stereogenic centers in aliskren















