Chapter 5: Q.22 (page 195)
If the two stereogenic centers of a compound are R,S in configuration, what are the R,S assignments for its enantiomer and two diastereomers?
Short Answer
R, R for enantiomers.
R, R and S, S for diastereomers.
Learning Materials
Features
Discover
Chapter 5: Q.22 (page 195)
If the two stereogenic centers of a compound are R,S in configuration, what are the R,S assignments for its enantiomer and two diastereomers?
R, R for enantiomers.
R, R and S, S for diastereomers.
All the tools & learning materials you need for study success - in one app.
Get started for free
How is compound A related to compounds B–E? Choose from enantiomers, diastereomers, constitutional isomers, or identical molecules.

What is the maximum number of stereoisomers possible for a compound with:
a. three stereogenic centers;
b. eight stereogenic centers?
Locate the tetrahedral stereogenic center(s) in each compound. A molecule may have one or more stereogenic centers.
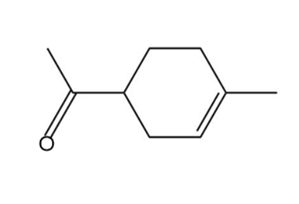
a.

b.
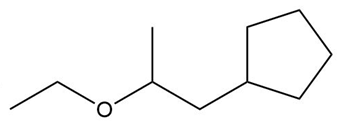
c.

d.
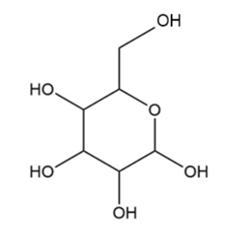
e.
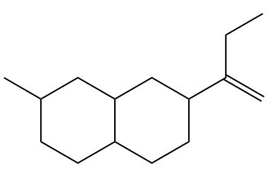
f.
Which of the following cyclic molecules are meso compounds?

a.

b.

c.
Rank the following groups in order of decreasing priority.
What do you think about this solution?
We value your feedback to improve our textbook solutions.