Chapter 5: Q.16 (page 190)
What is the maximum number of stereoisomers possible for a compound with:
a. three stereogenic centers;
b. eight stereogenic centers?
Short Answer
- 8 isomers.
- 256 isomers.
Learning Materials
Features
Discover
Chapter 5: Q.16 (page 190)
What is the maximum number of stereoisomers possible for a compound with:
a. three stereogenic centers;
b. eight stereogenic centers?
All the tools & learning materials you need for study success - in one app.
Get started for free
Drawn are four isomeric dimethylcyclopropanes.
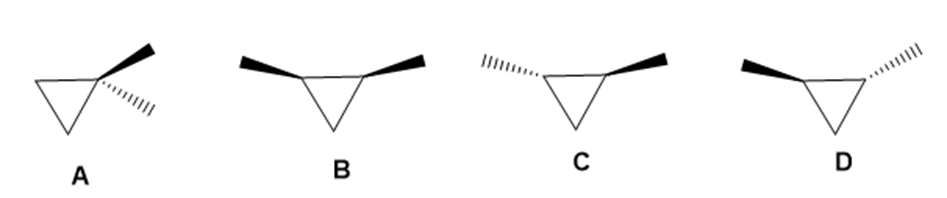
a. How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D?
b. Label each compound as chiral or achiral.
c. Which compounds, alone, would be optically active?
d. Which compounds have a plane of symmetry?
e. How do the boiling points of the compounds in each pair compare: A and B; B and C; C and D?
f. Which of the compounds are meso compounds?
g. Would an equal mixture of compounds C and D be optically active? What about an equal mixture of B and C?
The [α] of pure quinine, an antimalarial drug, is –165.
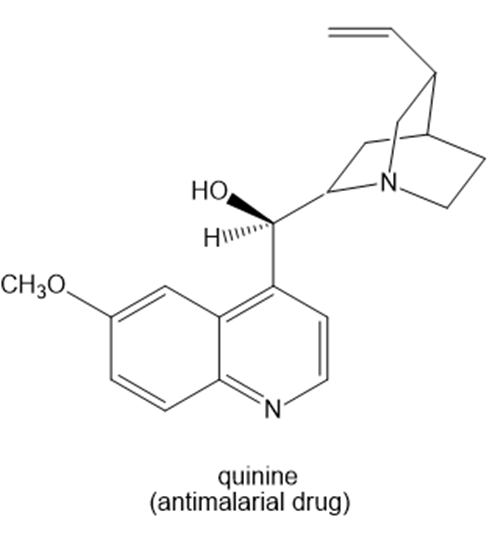
a. Calculate the ee of a solution with the following [α] values: –50, –83, and –120.
b. For each ee, calculate the percent of each enantiomer present.
c. What is [α] for the enantiomer of quinine?
d. If a solution contains 80% quinine and 20% of its enantiomer, what is the ee of the solution?
e. What is [α] for the solution described in part (d)?
Consider Newman projections (A–D) for four-carbon carbohydrates. How is each pairof compounds related: (a) A and B; (b) A and C; (c) A and D; (d) C and D? Choose fromidentical molecules, enantiomers, or diastereomers.
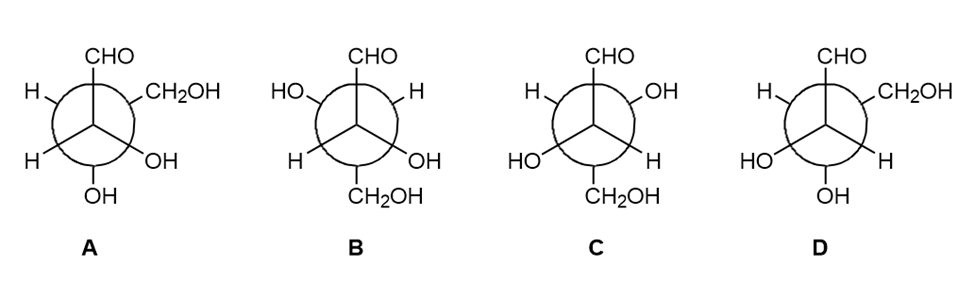
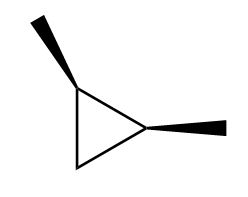
Draw in a plane of symmetry for each molecule.
a.
b.
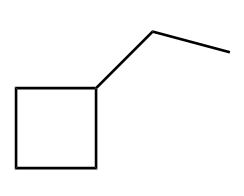
c.
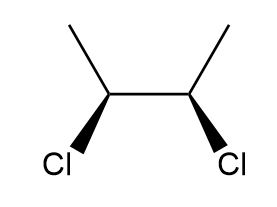
d.
Label the two stereogenic centers in each compound and draw all possible stereoisomers.
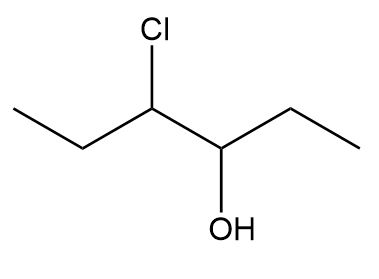
a.
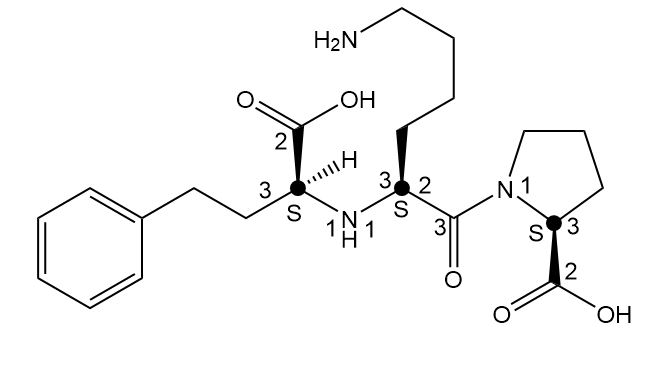
b.
What do you think about this solution?
We value your feedback to improve our textbook solutions.