Chapter 14: Q.21558-14-68P (page 567)
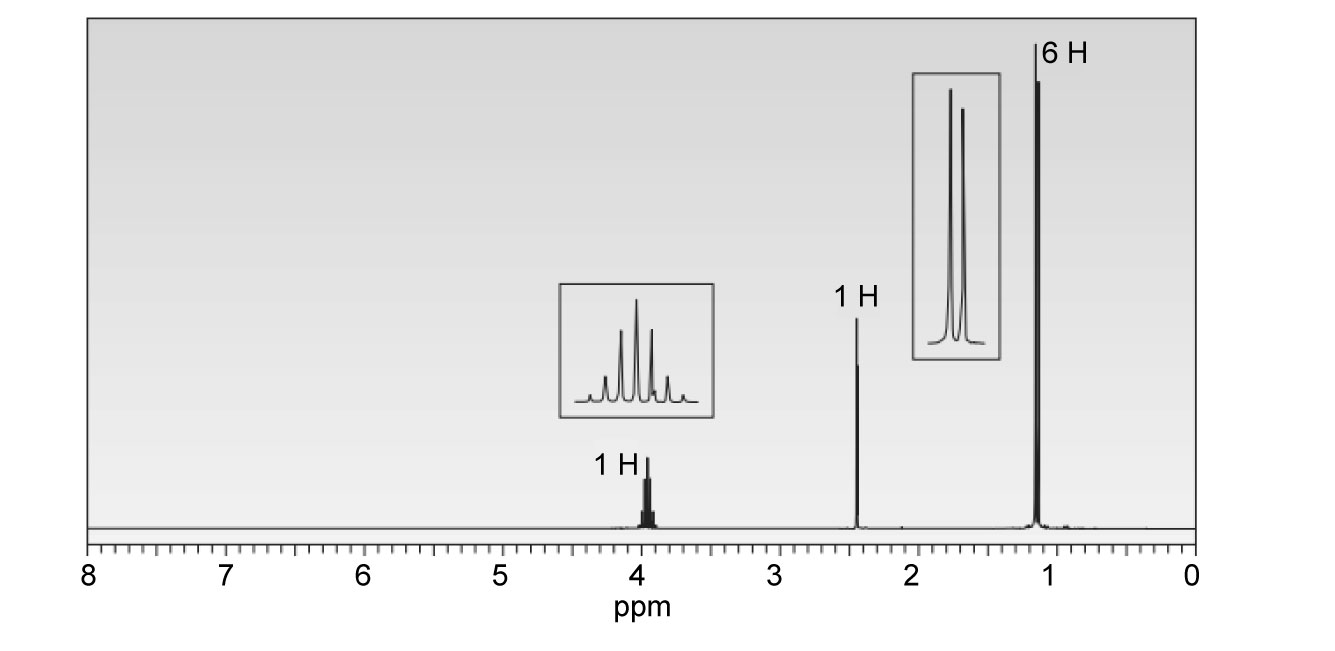
Question. Compound Q has molecular formula C5H9CIO2. Deduce the structure of P from its 1H and 13C -NMR spectra.
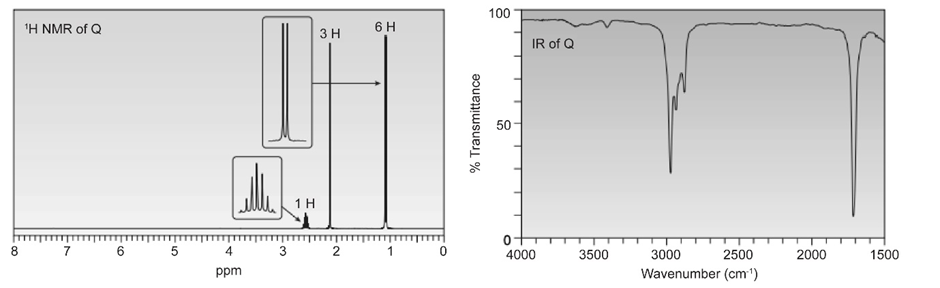
Short Answer
Answer
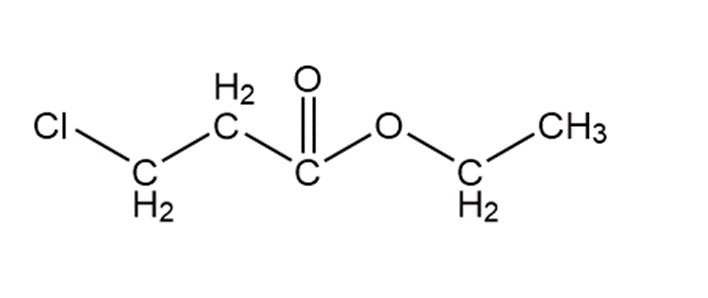
The structure of Q is as follows:
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-68P (page 567)
Question. Compound Q has molecular formula C5H9CIO2. Deduce the structure of P from its 1H and 13C -NMR spectra.

Answer
The structure of Q is as follows:

All the tools & learning materials you need for study success - in one app.
Get started for free
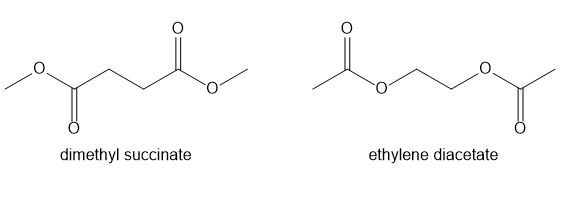
Compound A exhibits two signals in its 1H NMR spectrum at 2.64 and 3.69 ppm and the ratio of the absorbing signals is 2:3. Compound B exhibits two signals in its 1H NMR spectrum at 2.09 and 4.27 ppm and the ratio of the absorbing signals is 3:2. Which compound corresponds to dimethyl succinate and which compound corresponds to ethylene diacetate?
Propose a structure for a compound of molecular formula C3H8O with an IR absorption at 3600 to 3200 cm-1 and the following NMR spectrum:
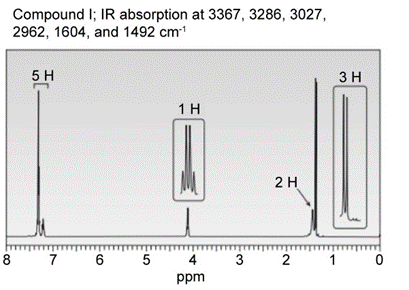
Question: Identify the structures of isomers H and I (molecular form C8H11N)
a.
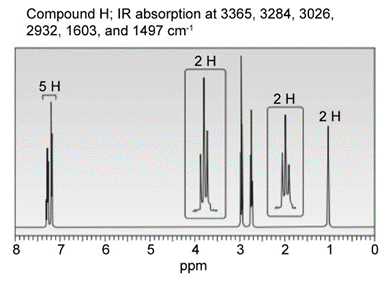
b.
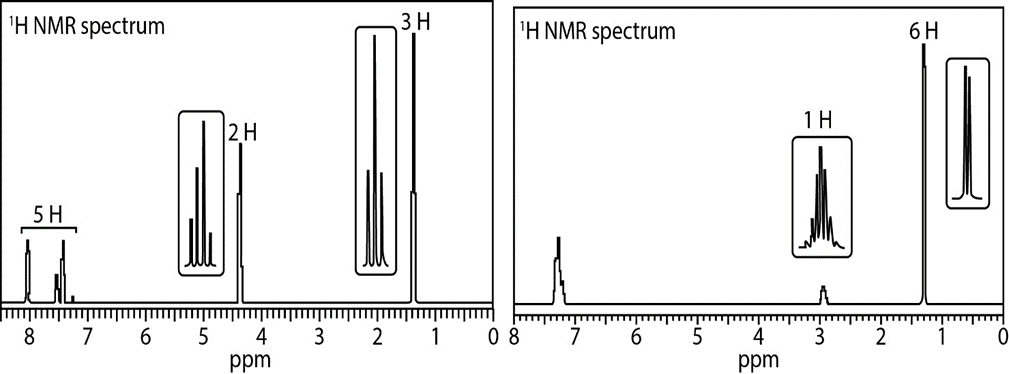
Propose a structure consistent with each set of data.
a. C9H10O2: IR absorption at 1718cm-1
b. C9H12 : IR absorption at 2850–3150cm-1
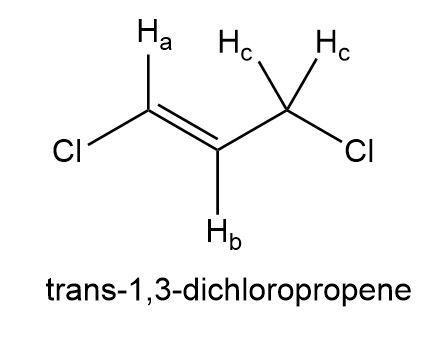
Draw a splitting diagram for Hb in trans-1,3-dichloropropene, given that Jab = 13.1 Hz and Jbc = 7.2 Hz.
What do you think about this solution?
We value your feedback to improve our textbook solutions.