Chapter 14: Q.21558-14-19P. (page 551)
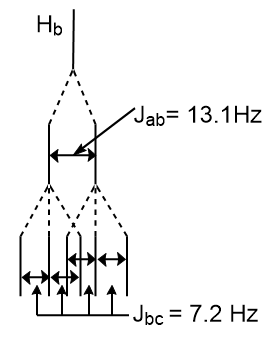
Draw a splitting diagram for Hb in trans-1,3-dichloropropene, given that Jab = 13.1 Hz and Jbc = 7.2 Hz.
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-19P. (page 551)
Draw a splitting diagram for Hb in trans-1,3-dichloropropene, given that Jab = 13.1 Hz and Jbc = 7.2 Hz.


All the tools & learning materials you need for study success - in one app.
Get started for free
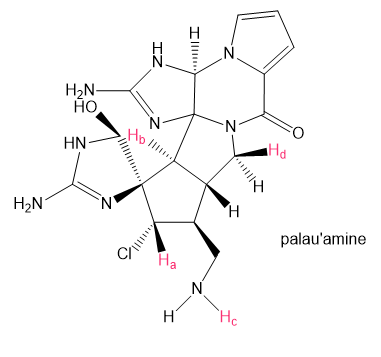
How many peaks are observed in the 1HNMR signal for each proton shown in red in palau'amine, the complex chapter-opening molecule?
Draw all constitutional isomers of molecular formula \({{\bf{C}}_{\bf{3}}}{{\bf{H}}_{\bf{6}}}{\bf{C}}{{\bf{l}}_{\bf{2}}}\).
a. How many signals does each isomer exhibit in its \({}^{\bf{1}}{\bf{H}}\) NMR spectrum?
b. How many lines does each isomer exhibit in its \({}^{{\bf{13}}}{\bf{C}}\) NMR spectrum?
c. When only the number of signals in both \({}^{\bf{1}}{\bf{H}}\) and \({}^{{\bf{13}}}{\bf{C}}\) NMR spectroscopy is considered, is it possible to distinguish all of these constitutional isomers?
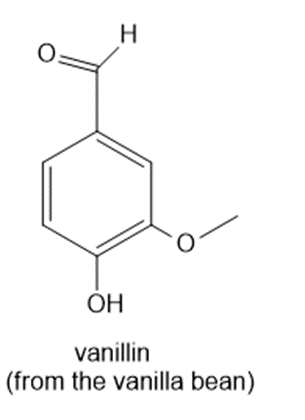
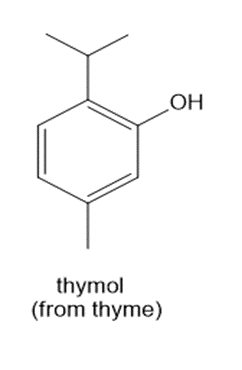
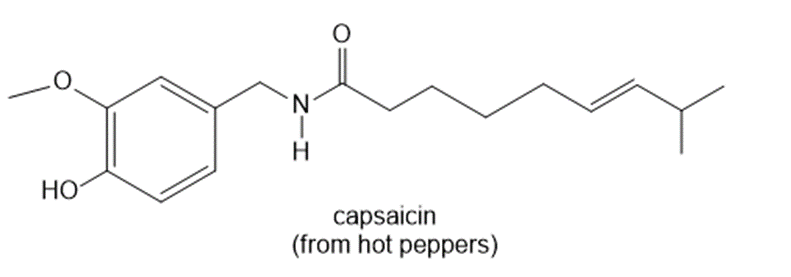
Question: How many 1H NMR signals does each natural product exhibit?
a.
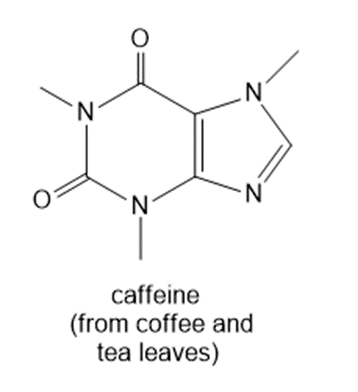
b.
c.
d.
Question. When 2-bromo-3,3-dimethylbutane is treated with K+- OC(CH3)3, a single product T having molecular formula C6H12 is formed. When 3,3-dimethylbutan-2-ol is treated with H2SO4 , the major product U has the same molecular formula. Given the following -NMR data, what are the structures of T and U? Explain in detail the splitting patterns observed for the three split signals in T. 1 H NMR of T: 1.01 (singlet, 9 H), 4.82 (doublet of doublets, 1 H, J = 10, 1.7 Hz), 4.93 (doublet of doublets, 1 H, J = 18, 1.7 Hz), and 5.83 (doublet of doublets, 1 H, J = 18, 10 Hz) ppm 1 H NMR of U: 1.60 (singlet) ppm.
Question: Reaction of C6H5CH2CH2OH with CH3COCl affords compound W, which has molecular formula C10H12O2. W shows prominent IR absorptions at 3088–2897, 1740, and 1606cm-1 . W exhibits the following signals in its 1 H NMR spectrum: 2.02 (singlet), 2.91 (triplet), 4.25 (triplet), and 7.20–7.35 (multiplet) ppm. What is the structure of W? We will learn about this reaction in Chapter 22.
What do you think about this solution?
We value your feedback to improve our textbook solutions.