Chapter 14: Q.21558-14-67P (page 567)
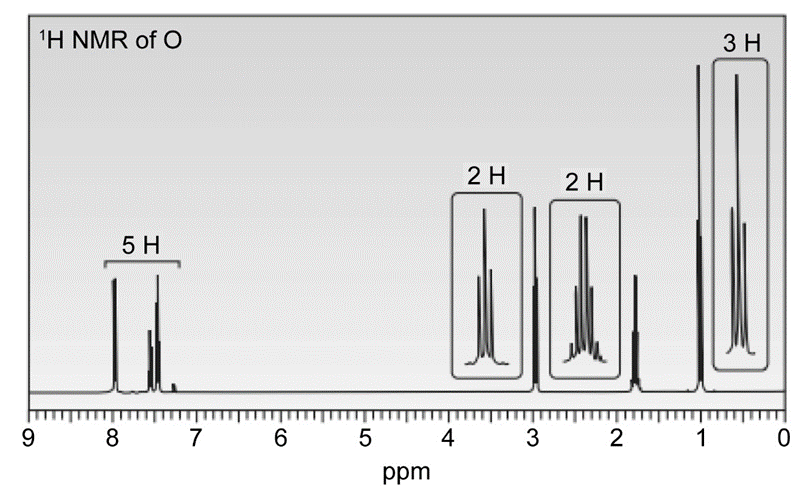
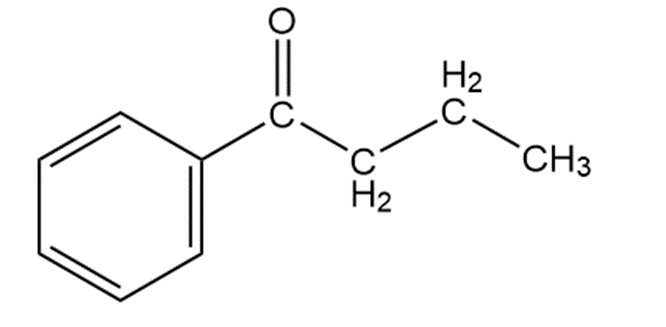
Question. Compound O has a molecular formula C10H12O and shows an IR absorption at 1687 . The 1H -NMR spectrum of O is given below. What is the structure of O?
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-67P (page 567)
Question. Compound O has a molecular formula C10H12O and shows an IR absorption at 1687 . The 1H -NMR spectrum of O is given below. What is the structure of O?

Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: How many 13C NMR signals do each compound exhibit?
Question: Identify the carbon atoms that give rise to each NMR signal.
a.
b
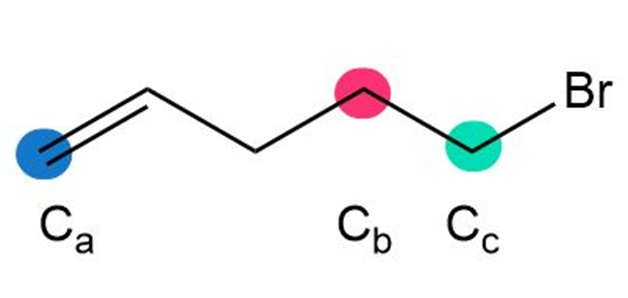
Question: Rank the highlighted carbon atoms in each compound in order of increasing chemical shift.
a.
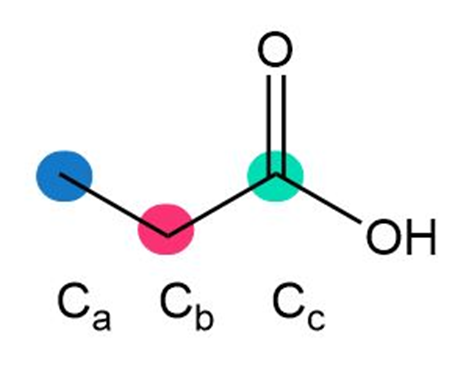
b.
Question: How many \(^{\bf{1}}{\bf{H}}\) NMR signals does each compound show?
a. 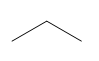
b. 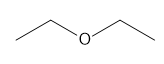
c. 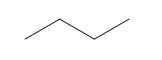
d. 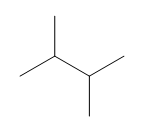
e. 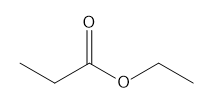
f. 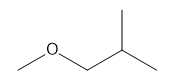
g. 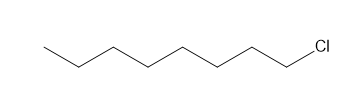
h. 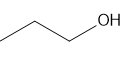
Label each statement as True or False.
a. When a nucleus is strongly shielded, the effective field is larger than the applied field and the absorption shifts downfield.
b. When a nucleus is strongly shielded, the effective field is smaller than the applied field and the absorption is shifted upfield.
c. A nucleus that is strongly deshielded requires a lower field strength for resonance.
d. A nucleus that is strongly shielded absorbs at a larger δ value.
What do you think about this solution?
We value your feedback to improve our textbook solutions.