Chapter 14: Q.21558-14-49P (page 563)
Question: Rank the highlighted carbon atoms in each compound in order of increasing chemical shift.
a.
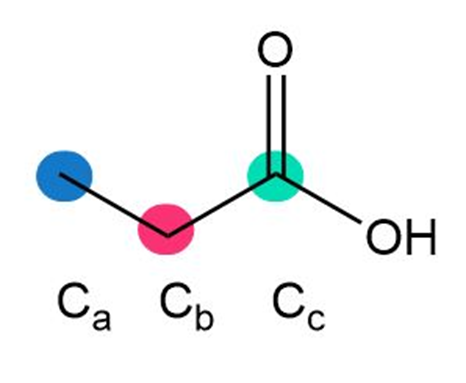
b.
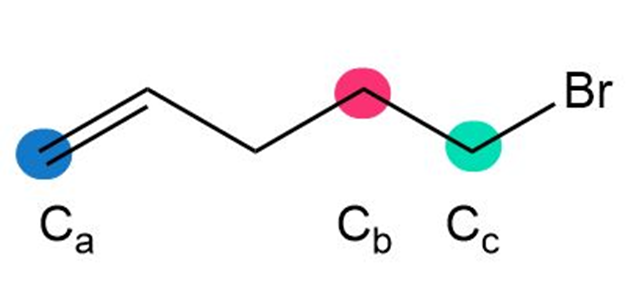
Short Answer
Answer
a. Ca< Cb < Cc
b. Cc < Cb< Ca
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-49P (page 563)
Question: Rank the highlighted carbon atoms in each compound in order of increasing chemical shift.
a.

b.

Answer
a. Ca< Cb < Cc
b. Cc < Cb< Ca
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: As we will learn in Chapter 20, reaction of (CH3)2CO with followed by affords compound D, which has a molecular ion in its mass spectrum at 84 and prominent absorptions in its IR spectrum at 3600–3200, 3303, 2938, and 2120cm-1 . D shows the following 1 H NMR spectral data: 1.53 (singlet, 6 H), 2.37 (singlet, 1 H), and 2.43 (singlet, 1 H) ppm. What is the structure of D?
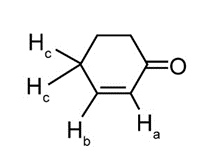
Question: Cyclohex-2-enone has two protons on its carbon-carbon double bond (labeled Ha and Hb ) and two protons on the carbon adjacent to the double bond (labeled Hc ). (a) If Jab = 11 Hz and Jbc = 4 Hz, sketch the splitting pattern observed for each proton on the hybridized carbons. (b) Despite the fact that Ha is located adjacent to an electron-withdrawing C=O, its absorption occurs up-field from the signal due to Hb(6.0 vs. 7.0 ppm). Offer an explanation.
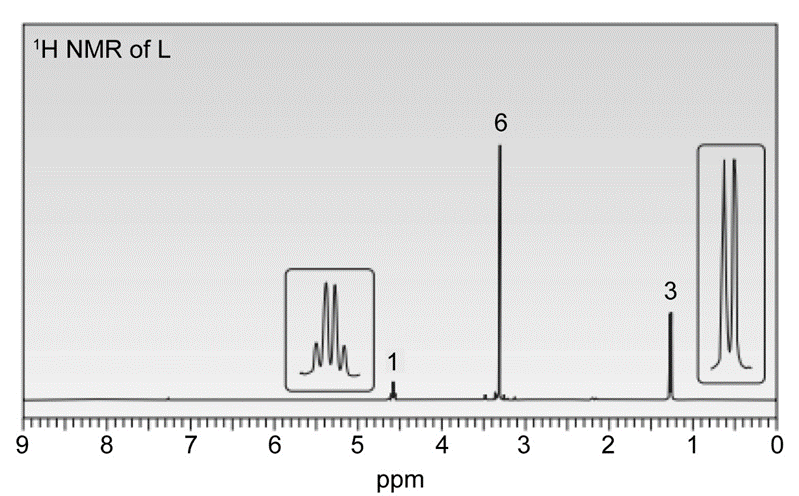
Question. In the presence of a small amount of acid, a solution of acetaldehyde(CH3CHO) in methanol (CH3OH) was allowed to stand and a new compound L was formed. L has a molecular ion in its mass spectrum at 90 and IR absorptions at 2992 and 2941cm-1 . L shows three signals in its 13C-NMR at 19, 52, and 101 ppm. 1H-NMR spectrum of L is given below. What is the structure of L?
Question. Treatment of (CH3)2 CHCH2CH(OH)CH2CH3 with TsOH affords two products (M and N) with the molecular formula C6H12 . The 1 H NMR spectra of M and N are given below. Propose structures for M and N, and draw a mechanism to explain their formation.
Question: How many \(^{\bf{1}}{\bf{H}}\) NMR signals does each dimethylcyclopropane show?
a. 
b. 
c.
What do you think about this solution?
We value your feedback to improve our textbook solutions.