Chapter 14: Q.21558-14-4P. (page 533)
How many 1H NMR signals does each dimethylcyclopropane show?
Short Answer
a. 2
b. 3
c. 3
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-4P. (page 533)
How many 1H NMR signals does each dimethylcyclopropane show?
a. 2
b. 3
c. 3
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: The \(^{\bf{1}}{\bf{H}}\) NMR spectrum 1,2-dimethoxyethane \(\left( {{\bf{C}}{{\bf{H}}_{\bf{3}}}{\bf{OC}}{{\bf{H}}_{\bf{2}}}{\bf{C}}{{\bf{H}}_{\bf{2}}}{\bf{OC}}{{\bf{H}}_{\bf{3}}}} \right)\) recorded on a 300 MHz NMR spectrometer consists of signals at 1017 Hz and 1065 Hz downfield from TMS. (a) Calculate the chemical shift of each absorption. (b) At what frequency would each absorption occur if the spectrum were recorded on a 500 MHz NMR spectrometer?
Question: a. How many signals does dimethyl fumarate ( CH3O2CCH=CHC02CH3 with a trans C=C) exhibit in its 13C NMR spectrum? b. Draw the structure of an isomer of dimethyl fumarate that has each of the following number of signals in its 13C NMR spectrum: [1] three; [2] four; [5] five
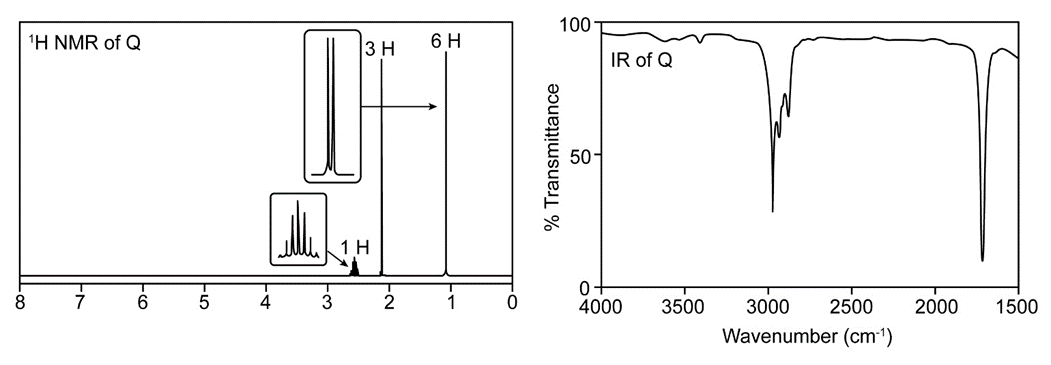
Question. Treatment of butan-2-one (CH3COCH2CH3) with a strong base followed by CH3l forms a compound Q, which gives a molecular ion in its mass spectrum at 86. The IR (> 1500 only) and 1H-NMR spectrum of Q is given below. What is the structure of Q?
Identify A and B, isomers of molecular formula C3H4Cl2 , from the given 1HNMR data: Compound A exhibits signals at 1.75 (doublet, 3H,J = 6.9 Hz) and 5.89 (quartet, 1H, J = 6.9 Hz) ppm. Compound B exhibits signals at 4.16 (singlet, 2 H), 5.42 (doublet, 1H, J = 1.9 Hz), and 5.59 (doublet, 1H, J = 1.9 Hz) ppm.
Rank each group of protons in order of increasing chemical shift.
a.
b.
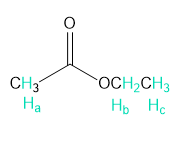
What do you think about this solution?
We value your feedback to improve our textbook solutions.