Chapter 14: Q.21558-14-11P. (page 541)
Rank each group of protons in order of increasing chemical shift.
a.
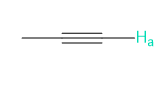
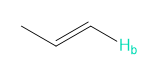
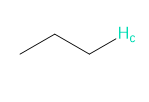
b.
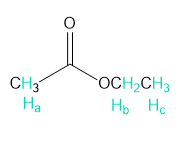
Short Answer
a.
b.
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-11P. (page 541)
Rank each group of protons in order of increasing chemical shift.
a.



b.

a.
b.
All the tools & learning materials you need for study success - in one app.
Get started for free
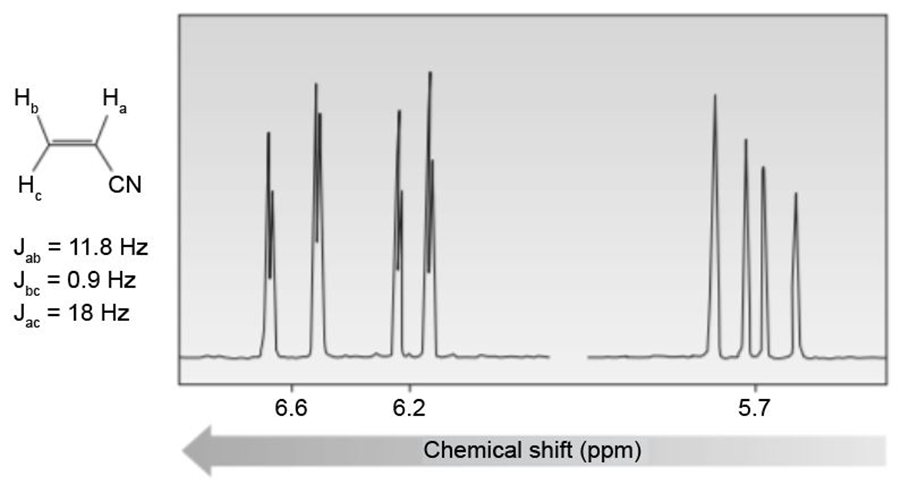
Question: Label the signals due to Ha ,Hb and Hc in the 1 H NMR spectrum of acrylonitrile (CH2=CHCN ). Draw a splitting diagram for the absorption due to the proton Hc.
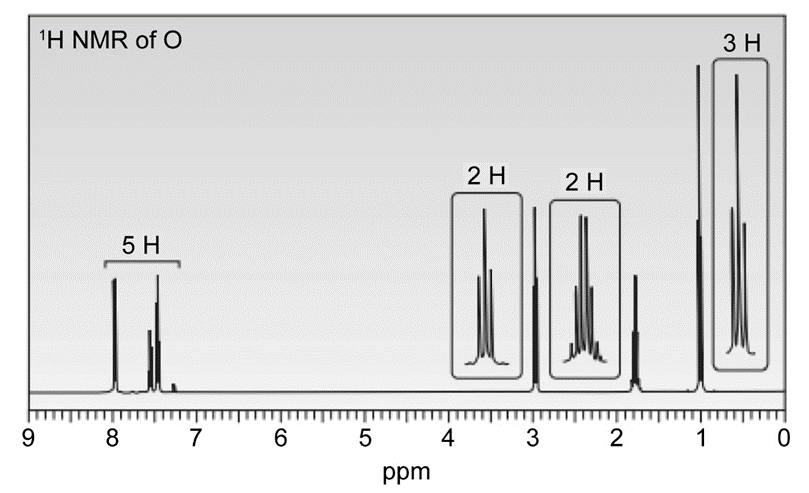
Question. Compound O has a molecular formula C10H12O and shows an IR absorption at 1687 . The 1H -NMR spectrum of O is given below. What is the structure of O?
Question: The 1 H NMR spectrum of N,N-dimethylformamide shows three singlets at 2.9, 3.0, and 8.0 ppm. Explain why the two groups are not equivalent to each other, thus giving rise to two NMR signals.
Question: How many \(^{\bf{1}}{\bf{H}}\) NMR signals does each compound give?
a. 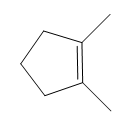
b. 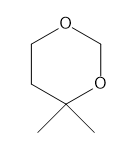
c.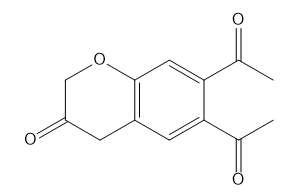
d. 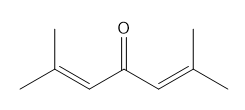
Question: The \(^{\bf{1}}{\bf{H}}\) NMR spectrum of \({\bf{C}}{{\bf{H}}_{\bf{3}}}{\bf{OH}}\) recorded on a 500 MHz NMR spectrometer consists of two signals, one due to the \({\bf{C}}{{\bf{H}}_{\bf{3}}}\) protons at 1715 Hz and one due to the OH proton at 1830 Hz, both measured downfield from TMS. (a) Calculate the chemical shift of each absorption. (b) Do the \({\bf{C}}{{\bf{H}}_{\bf{3}}}\) protons absorb upfield or downfield from the OH proton?
What do you think about this solution?
We value your feedback to improve our textbook solutions.