Chapter 14: Q.21558-14-51P (page 564)
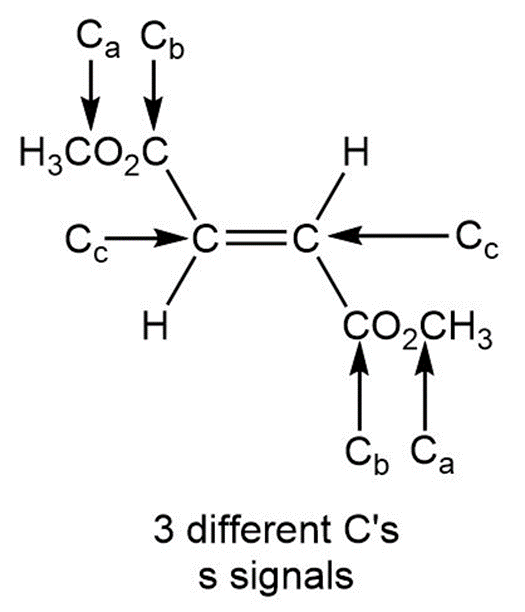
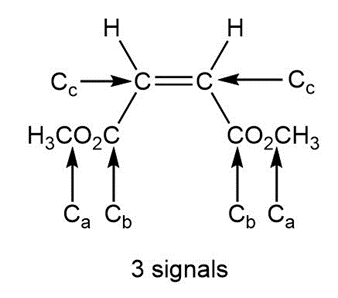
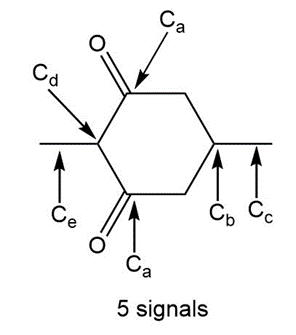
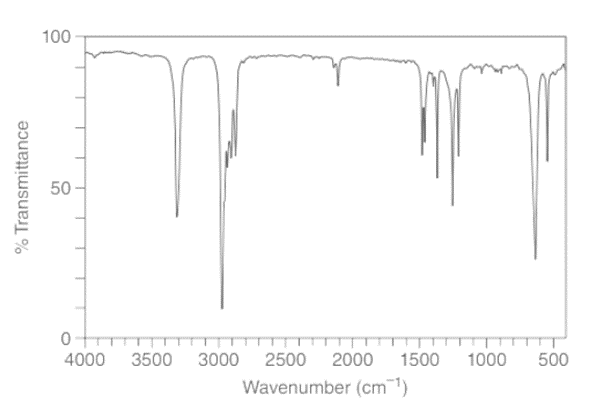
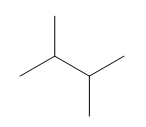
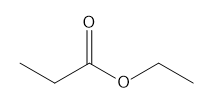
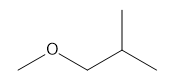
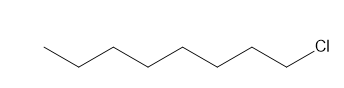
Question: a. How many signals does dimethyl fumarate ( CH3O2CCH=CHC02CH3 with a trans C=C) exhibit in its 13C NMR spectrum? b. Draw the structure of an isomer of dimethyl fumarate that has each of the following number of signals in its 13C NMR spectrum: [1] three; [2] four; [5] five
Short Answer
Answer
a.
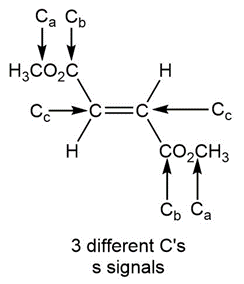
b.