Chapter 14: Q.21558-14-3P. (page 533)
How many 1H NMR signals does each compound show?
a.
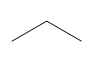
b.
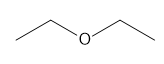
c.
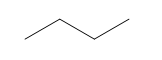
d.
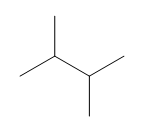
e.
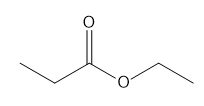
f.
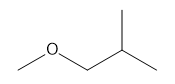
g.
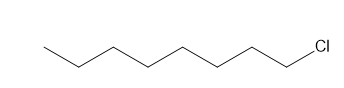
h.
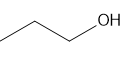
Short Answer
a. 2
b. 2
c. 2
d. 2
e. 4
f. 4
g. 5
h. 4
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-3P. (page 533)
How many 1H NMR signals does each compound show?
a.

b.

c.

d.

e.

f.

g.

h.

a. 2
b. 2
c. 2
d. 2
e. 4
f. 4
g. 5
h. 4
All the tools & learning materials you need for study success - in one app.
Get started for free
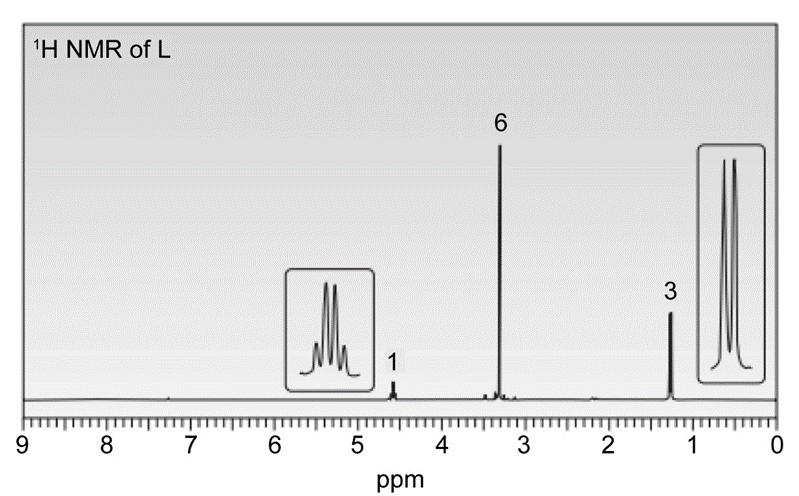
Question. In the presence of a small amount of acid, a solution of acetaldehyde(CH3CHO) in methanol (CH3OH) was allowed to stand and a new compound L was formed. L has a molecular ion in its mass spectrum at 90 and IR absorptions at 2992 and 2941cm-1 . L shows three signals in its 13C-NMR at 19, 52, and 101 ppm. 1H-NMR spectrum of L is given below. What is the structure of L?
Question: As we will learn in Chapter 20, reaction of (CH3)2CO with followed by affords compound D, which has a molecular ion in its mass spectrum at 84 and prominent absorptions in its IR spectrum at 3600–3200, 3303, 2938, and 2120cm-1 . D shows the following 1 H NMR spectral data: 1.53 (singlet, 6 H), 2.37 (singlet, 1 H), and 2.43 (singlet, 1 H) ppm. What is the structure of D?
Question: How could you use chemical shift and integration data in 1H NMR spectroscopy to distinguish between CH3OCH2CH2OCH3and CH3OCH2OCH3? The 1H NMR spectrum of each compound contains only singlets.
Question: Propose a structure consistent with each set of data.
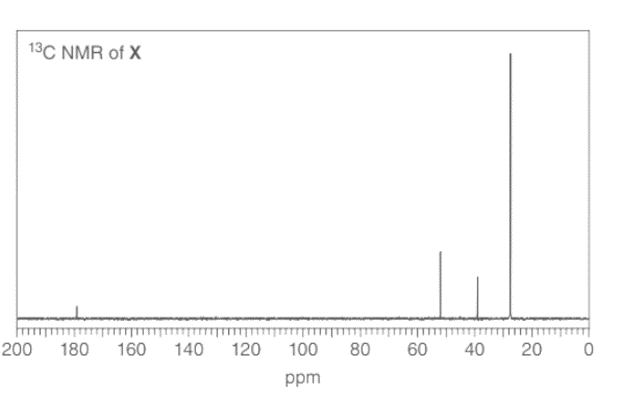
a. A compound X (molecular formula C6H12O2) gives a strong peak in its IR spectrum at 1740 cm-1. The 1 H NMR spectrum of X shows only two singlets, including one at 3.5 ppm. The 13C NMR spectrum is given below Propose a structure for X.
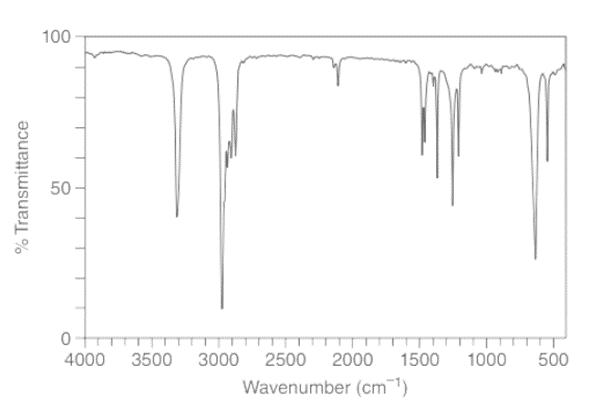
b. A compound Y (molecular formula C6H10 ) gives four lines in its 13C NMR spectrum (27, 30, 67, and 93 ppm), and the IR spectrum given here. Propose a structure for Y.
Question: (a) How many 1H NMR signals does each compound show? (b) Into how many peaks is each signal split?
What do you think about this solution?
We value your feedback to improve our textbook solutions.