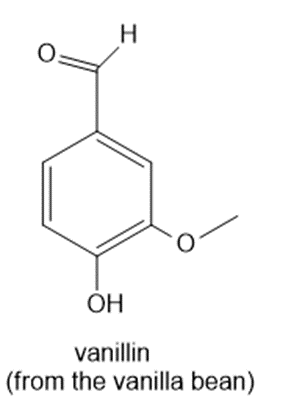
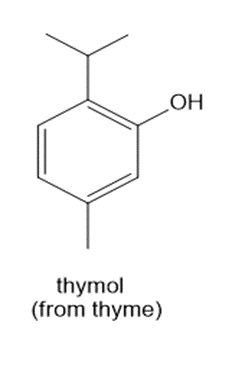
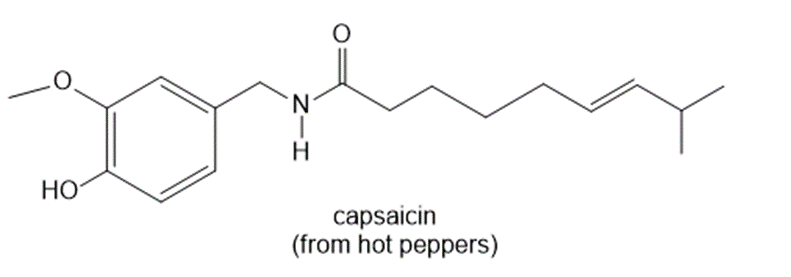
Chapter 14: Q.21558-14-20P. (page 551)
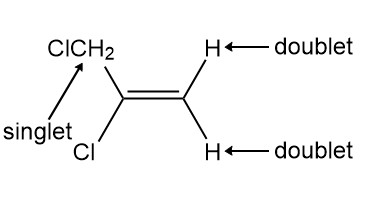
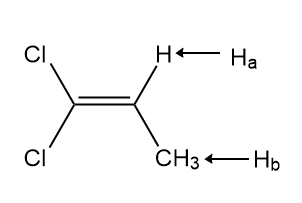
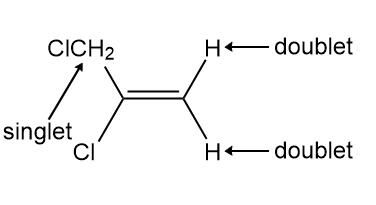
Identify A and B, isomers of molecular formula C3H4Cl2 , from the given 1HNMR data: Compound A exhibits signals at 1.75 (doublet, 3H,J = 6.9 Hz) and 5.89 (quartet, 1H, J = 6.9 Hz) ppm. Compound B exhibits signals at 4.16 (singlet, 2 H), 5.42 (doublet, 1H, J = 1.9 Hz), and 5.59 (doublet, 1H, J = 1.9 Hz) ppm.
Short Answer
A is 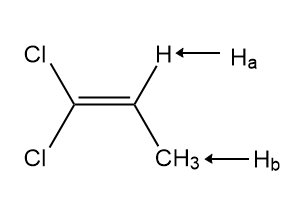
B is