Chapter 14: Q7P (page 527)
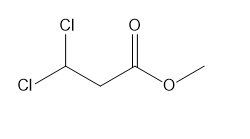
Question: How many \(^{\bf{1}}{\bf{H}}\) NMR signals would you expect for each compound?
a. 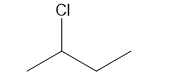
b. 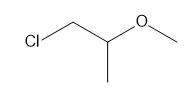
c.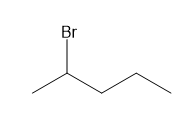
Short Answer
- 4
- 5
- 5
Learning Materials
Features
Discover
Chapter 14: Q7P (page 527)
Question: How many \(^{\bf{1}}{\bf{H}}\) NMR signals would you expect for each compound?
a. 
b. 
c.
All the tools & learning materials you need for study success - in one app.
Get started for free
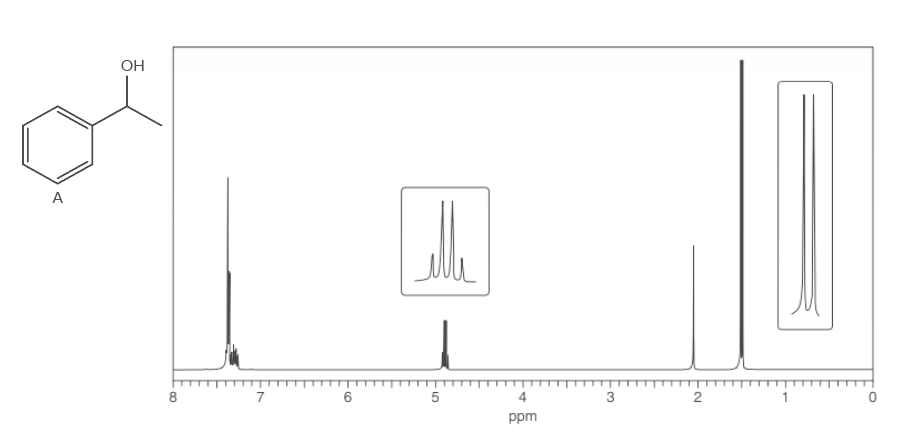
What protons in alcohol A give rise to each signal in its 1HNMR spectrum? Explain all splitting patterns observed for absorptions between 0–7 ppm.
Question. Compound Q has molecular formula C5H9CIO2. Deduce the structure of P from its 1H and 13C -NMR spectra.
Propose a structure for a compound of molecular formula C7H14O2 with an IR absorption at 1740 cm-1 and the following 1HNMR data:
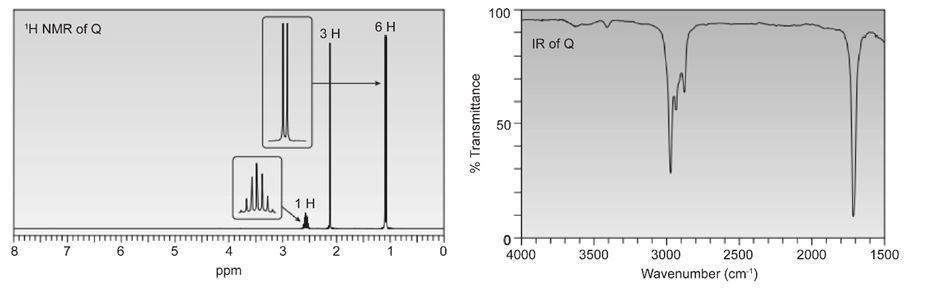
Which compounds give a 1H NMR spectrum with two signals in a ratio of 2:3?
a.
b.
c.
d.
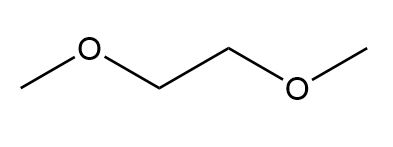
For each compound give the number of NMR signals, and then determine how many peaks are present for each NMR signal.
a.
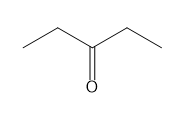
b.
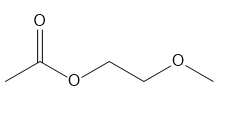
c.
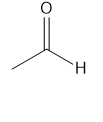
d.
What do you think about this solution?
We value your feedback to improve our textbook solutions.