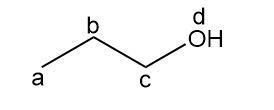
Chapter 14: Q.21558-14-21P. (page 552)
How many signals are present in the 1HNMR spectrum for each molecule? What splitting is observed in each signal?
a.
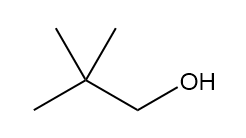
b.
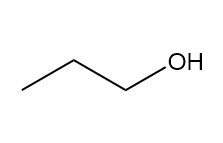
c.
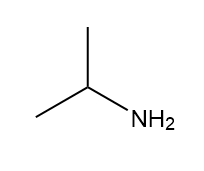
Short Answer
a.
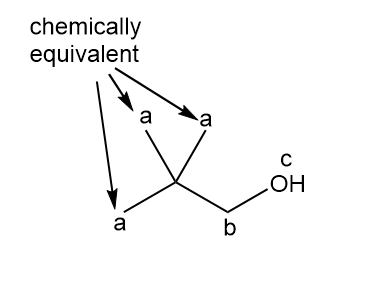
three signals (splitting observed: a - singlet; b-singlet; c-singlet).
b.
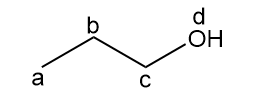
four signals (splitting observed: a - triplet; b-multiplet; c-triplet).
c.
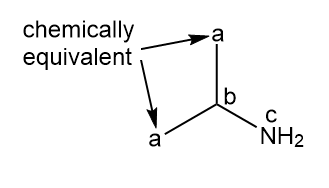
three signals (splitting observed: a - doublet; b-multiplet; c-singlet).