Chapter 14: Q.21558-14-63P (page 566)
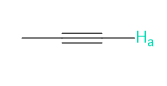
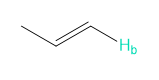
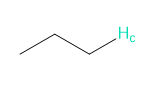
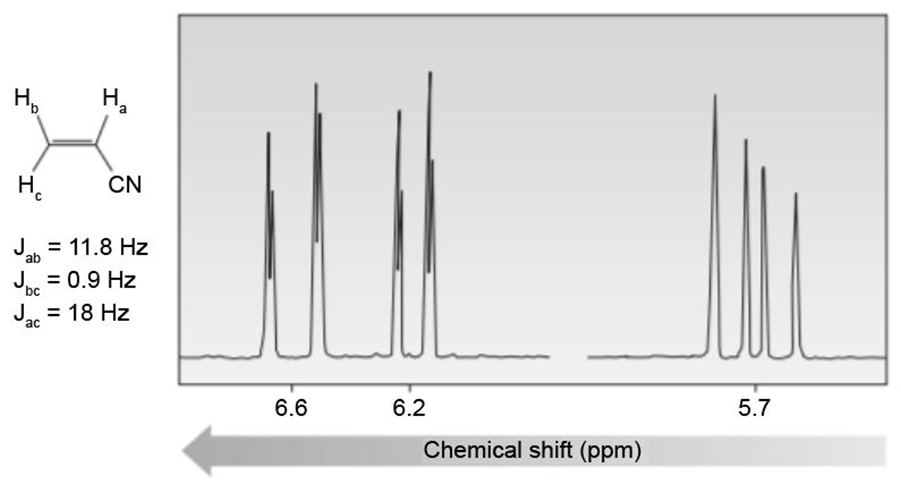
Question. Reaction of aldehyde D with amino alcohol E in the presence of NaH forms F (molecular formula C11H15NO2). F absorbs at 1730 cm-1in its IR spectrum. F also shows eight lines in its 13C-NMR spectrum, and gives the following -NMR spectrum: 2.32 (singlet, 6 H), 3.05 (triplet, 2 H), 4.20 (triplet, 2 H), 6.97 (doublet, 2 H), 7.82 (doublet, 2 H), and 9.97 (singlet, 1 H) ppm. Propose a structure for F. We will learn about this reaction in Chapter 18.
Short Answer
Answer
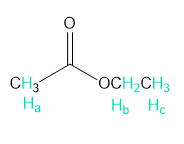
The structure of F is as follows:
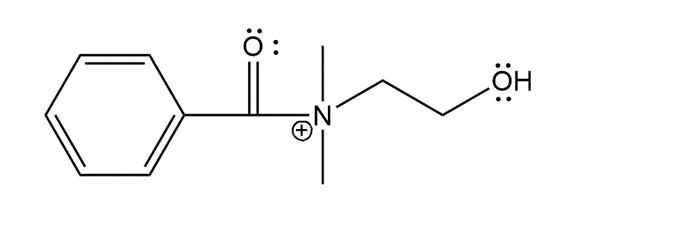
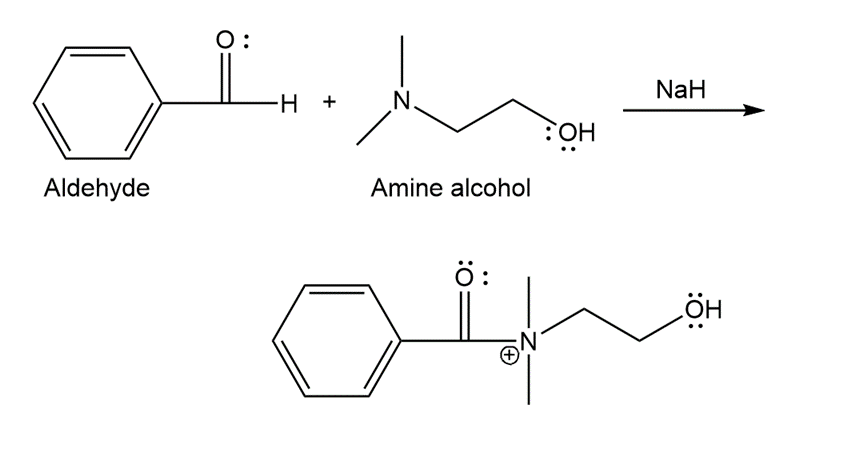
 Structure of F
Structure of F