Chapter 14: Q.21558-14-48P (page 563)
Question: How many 13C NMR signals do each compound exhibit?
Short Answer
Answer
a. Five
b. Three
c. Seven
d. Three
e. Seven
f. Five
g. Four
h. Three
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-48P (page 563)
Question: How many 13C NMR signals do each compound exhibit?
Answer
a. Five
b. Three
c. Seven
d. Three
e. Seven
f. Five
g. Four
h. Three
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Compound C has a molecular ion in its mass spectrum at 146 and a prominent absorption in its IR spectrum at 1762cm-1 . C shows the following 1H NMR spectral data: 1.47 (doublet, 3 H), 2.07 (singlet, 6 H), and 6.84 (quartet, 1 H) ppm. What is the structure of C?
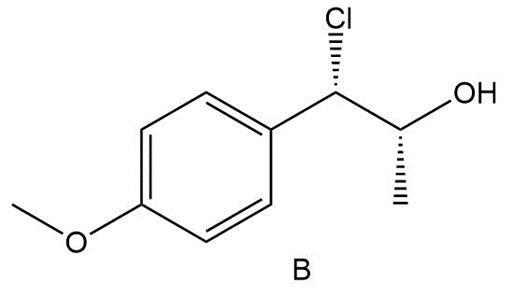
Question:Reaction of unknown A with HCl forms chlorohydrin B as the major product. A shows no absorptions in its IR spectrum at 1700 cm-1 or 3600-3200 cm-1 , and gives the following 1H NMR data: 1.4 (doublet, 3 H), 3.0 (quartet of doublets, 1 H), 3.5 (doublet, 1 H), 3.8 (singlet, 3 H), 6.9 (doublet, 2 H), and 7.2 (doublet, 2 H) ppm.
(a) Propose a structure for A, including stereochemistry.
(b) Explain why B is the major product in this reaction.
Question: How many \(^{\bf{1}}{\bf{H}}\) NMR signals does each dimethylcyclopropane show?
a. 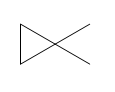
b. 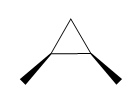
c.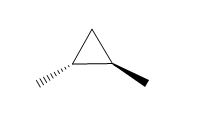
Question: Because 31P has an odd mass number, 31P nuclei absorb in the NMR and, in many ways, these nuclei behave similarly to protons in NMR spectroscopy. With this in mind, explain why the 1 H NMR spectrum of methyl dimethyl phosphonate,CH3PO(OCH3)2 , consists of two doublets at 1.5 and 3.7 ppm.
How many NMR signals does each compound give?
a.
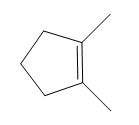
b.
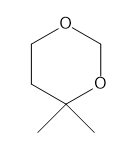
c.
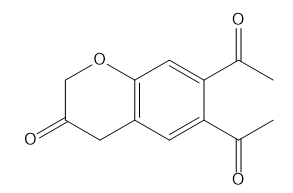
d.
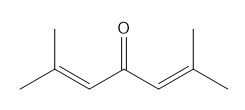
What do you think about this solution?
We value your feedback to improve our textbook solutions.