Chapter 14: Q.21558-14-61P (page 565)
Propose a structure consistent with each set of data.
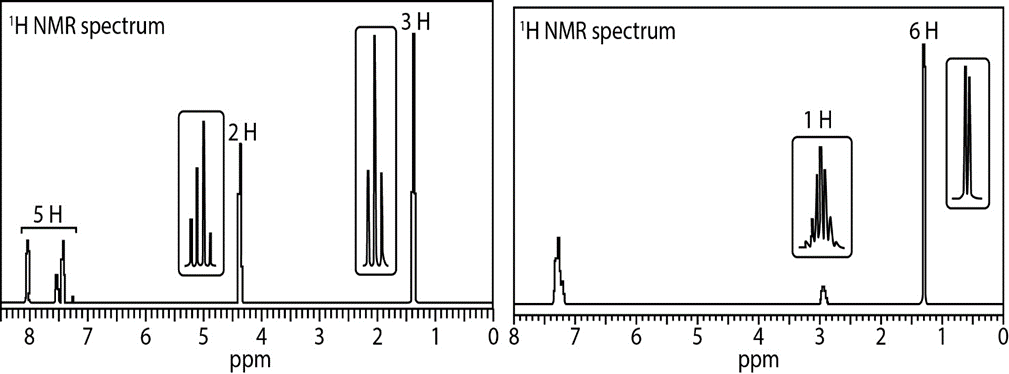
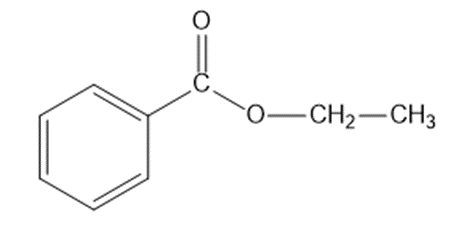
a. C9H10O2: IR absorption at 1718cm-1
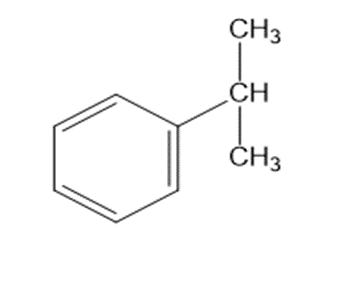
b. C9H12 : IR absorption at 2850–3150cm-1
Short Answer
Answer
a.
b.
Learning Materials
Features
Discover
Chapter 14: Q.21558-14-61P (page 565)
Propose a structure consistent with each set of data.
a. C9H10O2: IR absorption at 1718cm-1
b. C9H12 : IR absorption at 2850–3150cm-1

Answer
a.

b.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: How many different types of protons are present in each compound?
How many 1H NMR signals does each dimethylcyclopropane show?
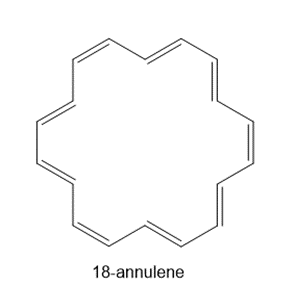
Question: 18-Annulene shows two signals in its 1 H NMR spectrum, one at 8.9 (12 H) and one at –1.8 (6 H) ppm. Using a similar argument to that offered for the chemical shift of benzene protons, explain why both shielded and deshielded values are observed for 18-annulene.
How many signals are present in the 1HNMR spectrum for each molecule? What splitting is observed in each signal?
a.
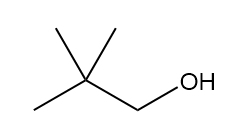
b.
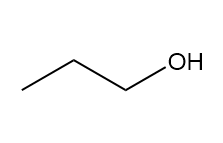
c.
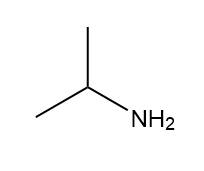
Question. When 2-bromo-3,3-dimethylbutane is treated with K+- OC(CH3)3, a single product T having molecular formula C6H12 is formed. When 3,3-dimethylbutan-2-ol is treated with H2SO4 , the major product U has the same molecular formula. Given the following -NMR data, what are the structures of T and U? Explain in detail the splitting patterns observed for the three split signals in T. 1 H NMR of T: 1.01 (singlet, 9 H), 4.82 (doublet of doublets, 1 H, J = 10, 1.7 Hz), 4.93 (doublet of doublets, 1 H, J = 18, 1.7 Hz), and 5.83 (doublet of doublets, 1 H, J = 18, 10 Hz) ppm 1 H NMR of U: 1.60 (singlet) ppm.
What do you think about this solution?
We value your feedback to improve our textbook solutions.