Chapter 22: Q84. (page 923)
Question. Draw a stepwise mechanism for the following reaction, the last step in a five-step industrial synthesis of vitamin C that begins with the simple carbohydrate glucose.
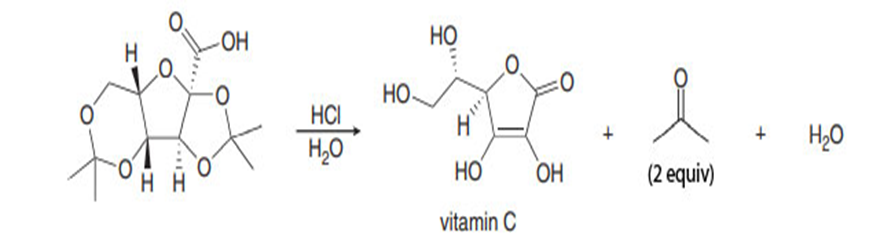
Short Answer
Answer
Step 1:
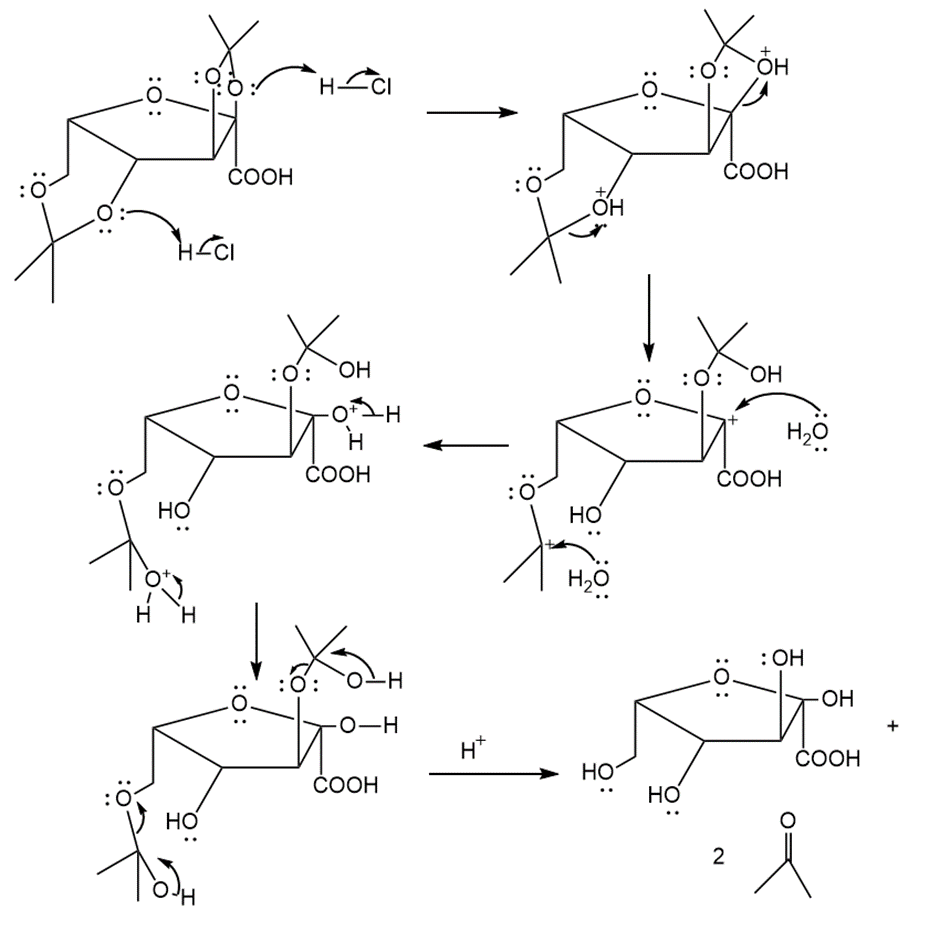
Step 2:
Learning Materials
Features
Discover
Chapter 22: Q84. (page 923)
Question. Draw a stepwise mechanism for the following reaction, the last step in a five-step industrial synthesis of vitamin C that begins with the simple carbohydrate glucose.

Answer
Step 1:

Step 2:
All the tools & learning materials you need for study success - in one app.
Get started for free
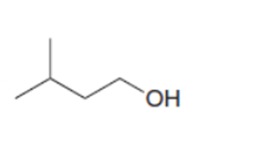
What reagent is needed to convert (CH3)2CHCH2COCIinto each compound?
a.
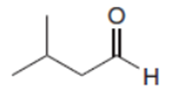
b.
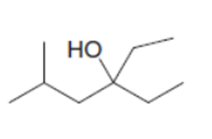
c.
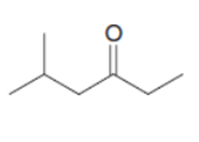
d.
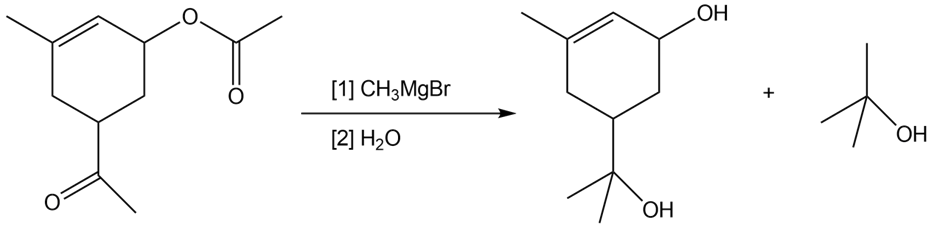
Draw a stepwise mechanism for the following reaction. Your mechanism must show how both organic products are formed.
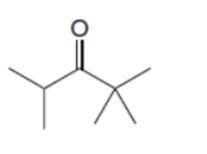
Draw two different ways to prepare each ketone from an acid chloride and an organocuprate reagent.
a.
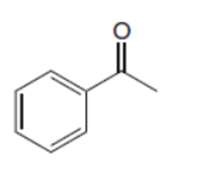
b.
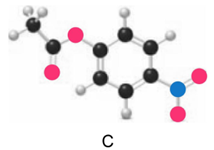
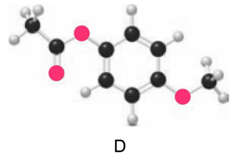
Question: Which ester, C or D, is more reactive in nucleophilic acyl substitution? Explain your reasoning.
Question: Draw the products of each reaction
a.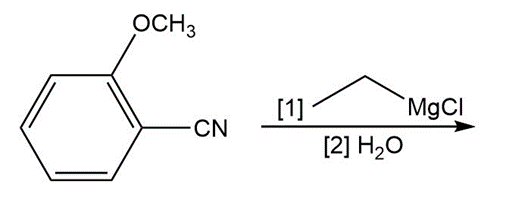
b. 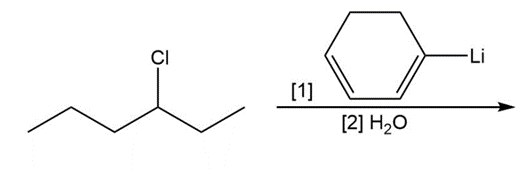
What do you think about this solution?
We value your feedback to improve our textbook solutions.