Chapter 22: Q39. (page 914)
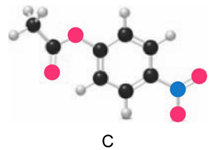
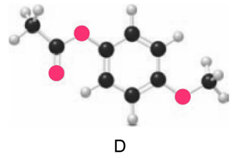
Question: Which ester, C or D, is more reactive in nucleophilic acyl substitution? Explain your reasoning.
Short Answer
Answer
Compound C is more reactive than compound D.
Learning Materials
Features
Discover
Chapter 22: Q39. (page 914)
Question: Which ester, C or D, is more reactive in nucleophilic acyl substitution? Explain your reasoning.


Answer
Compound C is more reactive than compound D.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Without reading ahead in Chapter 22, state whether it should be possible to carry out each of the following nucleophilic substitution reactions.
a.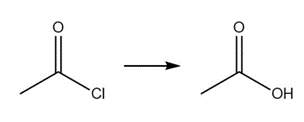
b.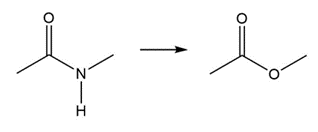
c.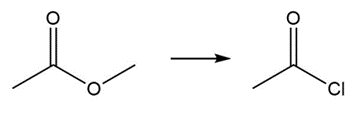
d.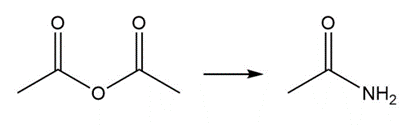
Question: Draw the products of each reaction.
a.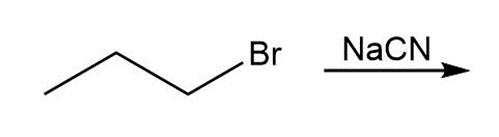
b. 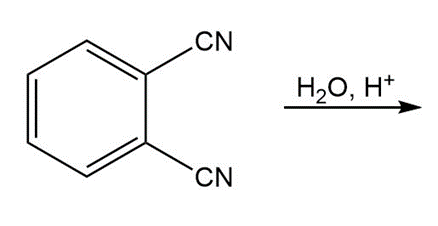
c. 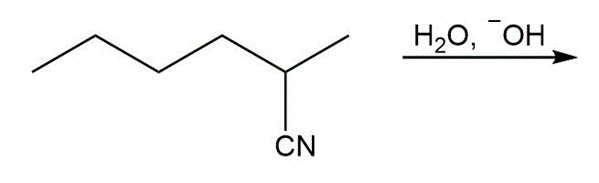
Question: Draw the products of each reaction.
a.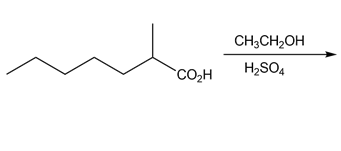
b. 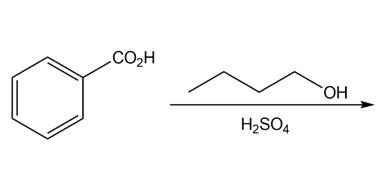
c.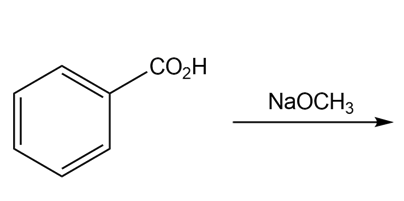
d. 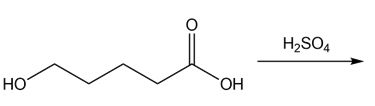
Explain why metal hydride reduction gives an endo alcohol as the major product in one reaction given below and an exo alcohol as the major product in the other reaction.
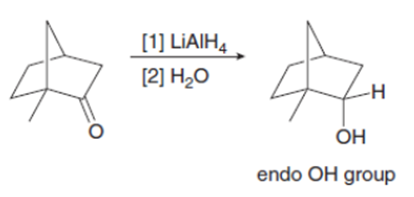
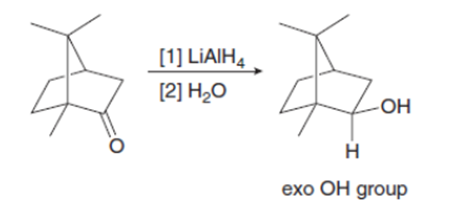
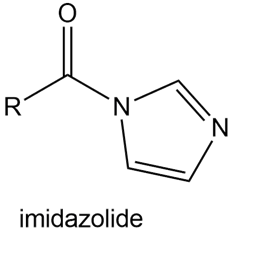
Question: Explain why imidazolides are much more reactive than other amides in nucleophilic acyl substitution.
What do you think about this solution?
We value your feedback to improve our textbook solutions.