Chapter 22: Q31. (page 908)
Question: Draw the products of each reaction.
a.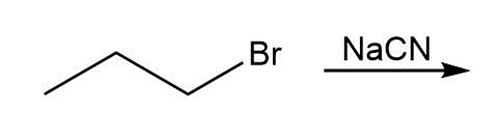
b. 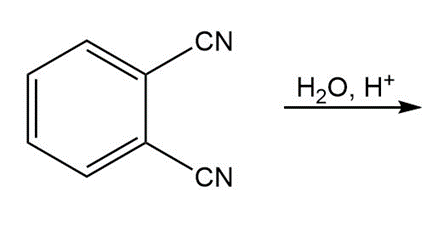
c. 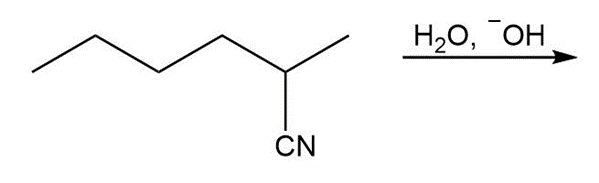
Short Answer
Answer
a.
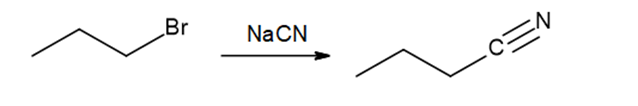
b.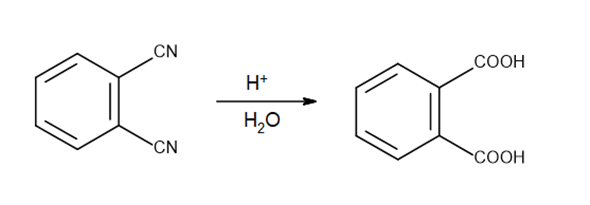
c.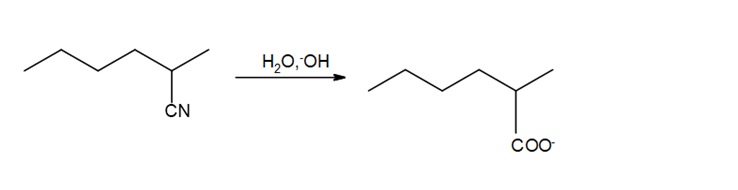
Learning Materials
Features
Discover
Chapter 22: Q31. (page 908)
Question: Draw the products of each reaction.
a.
b. 
c. 
Answer
a.

b.
c.
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: How do the following experimental results support the resonance description of the relative stability of acid chlorides compared to amides? The C-Cl bond lengths in and CH3COCl are identical (178 pm), but the C-N bond in is shorter than the C-N bond in (135 pm versus 147 pm).
What Grignard reagent and carbonyl compound are needed to prepare each alcohol? As shown in part (d), 3o alcohols with three different R groups on the carbon bonded to the OH group can be prepared by three different Grignard reactions.
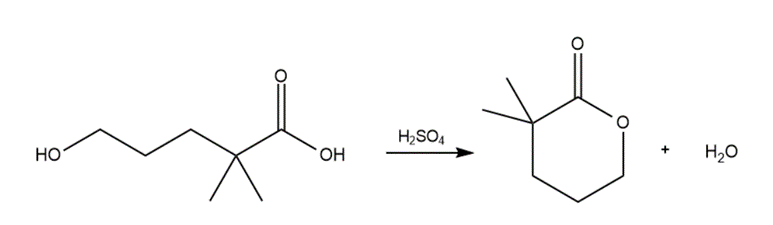
Draw a stepwise mechanism for the following reaction.
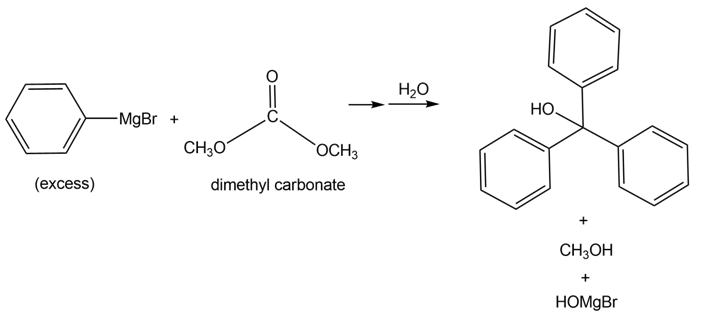
Tertiary alcohols can be formed by the reaction of dimethyl carbonate [(CH3O)2CO ] with excess Grignard reagent. Draw a stepwise mechanism for the following transformation.
A student tried to carry out the following reaction sequence, but none of diol A was formed. Explain what was wrong with this plan, and design a successful stepwise synthesis of A.
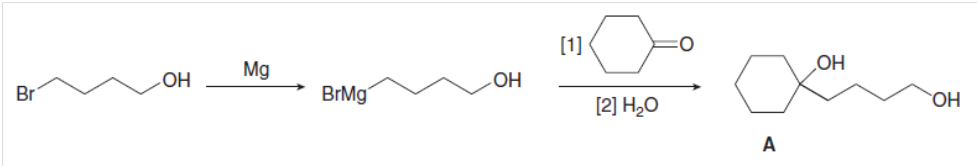
What do you think about this solution?
We value your feedback to improve our textbook solutions.